The Stille reaction is a member of a large family of palladium-catalysed cross-coupling reactions. However, its generality and high degree of success have made it popular among synthetic chemists. One obvious reason for such popularity is the reactions versatility. It is frequently used in the total syntheses of large polyfunctional molecules for the coupling of complex subunits. However, it is sometimes found that it is not universally applicable and that many substrates require specific reaction conditions for cross-coupling to be successful. There are relatively few examples of the successful introduction of alkyl groups employing Stille cross-coupling reactions described in the literature, and for alpha-iodocyclohexenones the reported yields are only moderate. This is probably due to failure resulting from competing beta-hydride elimination when employing stannanes with alkyl groups bearing beta-hydrogens and to the low reactivity of the system. A further problem often encountered is the competing reduction of vinylic or aromatic halides to the corresponding hydrocarbon. This can be a serious problem where the halide is unreactive.
Example:
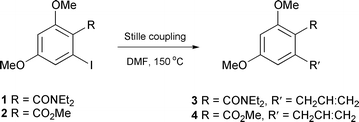
Nevertheless this seems like a very versatile (although rather unexplored) reaction in combinatory chemistry.
Synthesis of functionalized styrenes via palladium-catalyzed coupling of aryl bromides with vinyl tin reagents (D. R. McKean, G. Parrinello, A. F. Renaldo, J. K. Stille)J. Org. Chem. (1987) 52(3) (422-424)
abstract
Highly functionalized styrene derivatives have been synthesized in a single step by the palladium-catalysed coupling of aryl bromides with tributylethenylstannate. Aryl bromides substituted with electron-withdrawing groups couple rapidly under the the reaction conditions, while aryl bromides containing electron-donating substituents require further addition of catalyst for complete conversion. 1,4-Dibromobenzene can be coupled in a highly selective fashion with either 1 or 2 equiv of tin reagent to give 4-bromostyrene or diethehnylbenzene, respectively.
The effect of diethylamine on Stille alkylations with tetraalkylstannanes(M. Teresa Barros, Christopher D. Maycock, Mariana I. Madureira and M. Rita Ventura)Chem. Commun., 2001, (17), 1662 - 1663 DOI:
10.1039/b103072h

Abstract
The addition of diethylamine to Stille alkylation reactions using stannanes improves yields by reducing beta-hydride elimination and reduction reactions, it also serves as a substitute for other additives such as Cu(i)I.
Mechanism of the Stille Reaction(Casado, A. L.; Espinet, P.)J. Am. Chem. Soc. 1998, 120(35), (8978-8985)DOI:
10.1021/ja9742388

abstract
The so far accepted mechanism of the Stille reaction (palladium-catalyzed cross-coupling of organotin reagents with organic electrophiles) is criticized. Based on kinetic studies on catalytic reactions, and on reactions with isolated intermediates, a corrected mechanism is proposed. The couplings between R1I (1) (R1 = C6Cl2F3 = 3,5-dichlorotrifluorophenyl) and R2SnBu3 (R2 = CH=CH2, 2a; C6H4-4-OCH3, 2b), catalyzed by trans-[PdR1I(AsPh3)2] (3a), give R1-R2 and obey a first-order law, robs = a[3a][2a]/(b + [AsPh3]), with a = (2.31 ± 0.09) × 10-5 s-1 and b = (6.9 ± 0.3) × 10-4 mol L-1, for [1] = [2a] = 0-0.2 mol L-1, [3a] = 0-0.02 mol L-1, and [AsPh3] = 0-0.07 mol L-1, at 322.6 K in THF. The only organopalladium(II) intermediate detected under catalytic conditions is 3a. The apparent activation parameters found for the coupling of 1 with 2a support an associative transmetalation step (deltaH(+-)obs = 50 ± 2 kJ mol-1, deltaS(+-)obs = -155 ± 7 J K-1 mol-1 in THF; and deltaH(+-)obs = 70.0 ± 1.7 kJ mol-1, deltaS(+-)obs = -104 ± 6 J K-1 mol-1 in chlorobenzene, with [1]0 = [2]0 = 0.2 mol L-1, [3a] = 0.01 mol L-1). The reactions of 2a with isolated trans-[PdR1X(AsPh3)2] (X = halide) show rates Cl > Br > I. From these observations, the following mechanism is proposed: Oxidative addition of R1X to PdLn gives cis-[PdR1XL2], which isomerizes rapidly to trans-[PdR1XL2]. This trans complex reacts with the organotin compound following a SE2(cyclic) mechanism, with release of AsPh3 (which explains the retarding effect of the addition of L), to give a bridged intermediate [PdR1L(mu-X)(mu-R2)SnBu3]. In other words, an L-for-R2 substitution on the palladium leads R2 and R1 to mutually cis positions. From there the elimination of XSnBu3 yields a three-coordinate species cis-[PdR1R2L], which readily gives the coupling product R1-R2. indole_amine