I don't understand Recemization, or is inversion the same thing, and when does it happen in the reduction of (psudo)ephedrine to Meth. I've always known that in order to end up with D(+) Methamphetamine , one had to start with L-(-)(Pseudo) ephedrine....
Now that I've been chasing the Phenylalanine route to Amphetamine for a while , I run into a question that i think I have no answer for. Able to acquire some R- Phenylalaninol, to run the RP/I reduction method , I'm asking myself if there will be some inversion ,i.e. (+) ------->(-) in the reaction , and the same for running the Phenylalaninol-------> chlorinated phenylalaninol-------> Hydrogenation with (Pd). I have read somewhere that Pd causes inversion.......will have to re-read that I guess.
I don't seem to have too much of a handle on this , I have read this article .....
Post 499052
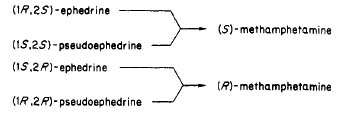
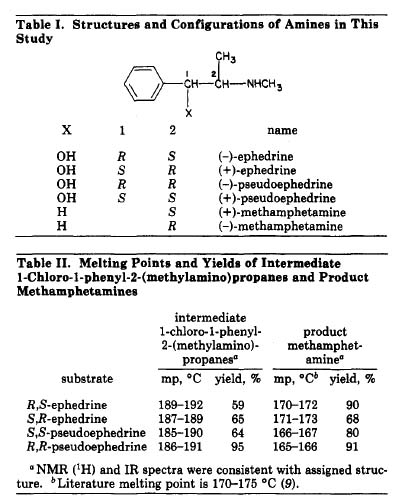
(Rhodium: "Meth Synthesis via Chloroephedrine", Stimulants)..........and the sorting of the 1S,1R,2S,2R, their equivelancy in the (+),(-) isomer , so now I'm really mixed up the graph in the article
Rhodium provided helps me understand at least the conditions of each carbon atom.
But my question remains will there be a change in my project , (+)-Phenylalaninol will it go from a (+) to (-) or will it remain the same. I'm taking a leap in assuming the (-)=L, and (+0)=R , after going through either the RP/I reaction or the Phenylalaninol---->chlorinated Phenylalaninol----> amphetamine.
Also, in general terms question, then during the process of reduction (Pseudo)ephedrine to methamphetamine via hydrogenation with Pd, where does the inversion occur in the process, and what is reaction is the culprit?........java

 Note
Note edited by java, all this reminds me or sine wave addition, with amplitude and direction, with occasional disruptions by discharging gates caused by high ionic solutions.