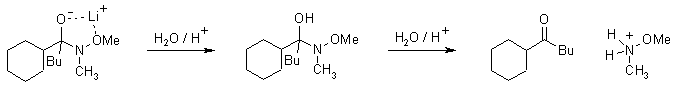Grignard Reaction / Grignard Reagents
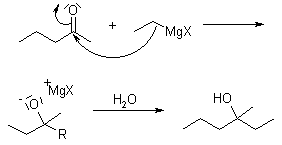
The Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or aldehyde, to form a tertiary or secondary alcohol, respectively. The reaction with formaldehyde leads to a primary alcohol.
Grignard Reagents are also used in the following important reactions: The addition of an excess of a Grignard reagent to an ester or lactone gives a tertiary alcohol in which two alkyl groups are the same, and the addition of a Grignard reagent to a nitrile produces an unsymmetrical ketone via a metalloimine intermediate. (Some more reactions are depicted below)
Mechanism
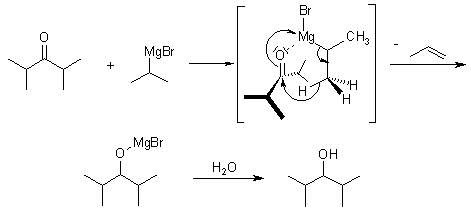
While the reaction is generally thought to proceed through a nucleophilic addition mechanism, sterically hindered substrates may react according to an SET (single electron transfer) mechanism:

With sterically hindered ketones the following side products are received:
The Grignard reagent can act as base, with deprotonation yielding an enolate intermediate. After work up, the starting ketone is recovered.

A reduction can also take place, in which a hydride is delivered from the ß-carbon of the Grignard reagent to the carbonyl carbon via a cyclic six-membered transition state.
Additional reactions of Grignard Reagents:
With carboxylic acid chlorides:
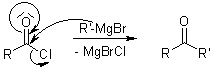
Esters are less reactive than the intermediate ketones, therefore the reaction is only suitable for synthesis of tertiary alcohols using an excess of Grignard Reagent:

With nitriles:

With CO2 (by adding dry ice to the reaction mixture):
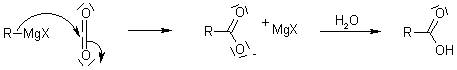
With oxiranes:
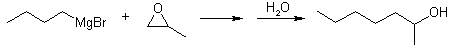
Recent Literature
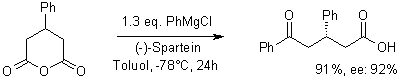
R. Shintani, G. C. Fu, Angewandte Chemie, 2002, 114, 1099.
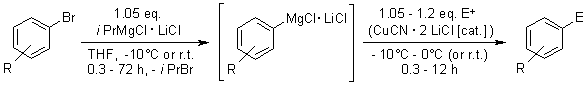
A. Krasovski, P. Knochel, Angew. Chem., 2004, 116, 3396-3399.
Abstract
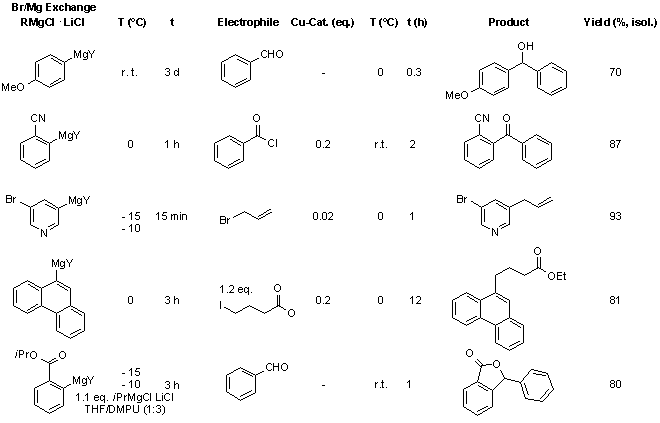
iPrMgCl·LiCl can be used for the simple, high-yielding preparation of a broad range of functionalized aryl- and heteroarylmagnesium reagents from aryl bromides. All reactions proceed within a convenient range of temperatures (-15°C - r.t.) and can be extended to the large-scale preparation of Grignard reagents.
Zusammenfassung
iPrMgCl·LiCl ist ein nützliches Reagenz für die Darstellung einer grossen Bandbreite an (Hetero-)Arylmagnesium-Verbindungen aus Arylbromiden in sehr guten Ausbeuten. Die Reaktionen verlaufen innerhalb eines gut zugänglichen Temperaturbereiches (-15°C bis Raumtemperatur) und können problemlos für den grossen Massstab angepasst werden
Related Reactions:
Nozaki-Hiyama Coupling / Nozaki-Hiyama-Kishi Reaction
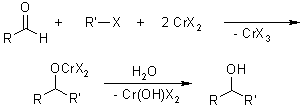
This coupling between halides and aldehydes is a chromium-induced redox reaction. Key A key advantage is the high chemoselectivity toward aldehydes. A disadvantage is the use of excess toxic chromium salts.
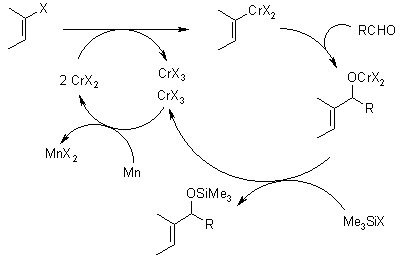
Newer methods allow the use of catalytic amounts chromium(II), which is regenerated by reduction with manganese or via electrochemical reduction.
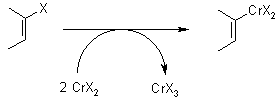
Mechanism

Catalyzed Reaction:
Recent Literature
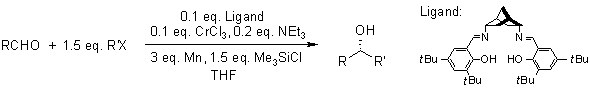
A. Berkessel, D. Menche, C. A. Sklorz, M. Schroeder, I. Paterson, Angew. Chem., 2003, 115, 1062-1065.

M. Bandini, P. G. Cozzi, P. Melchiorre, S. Morganti, A. Umani-Ronchi, Org. Lett., 2001, 3, 1153-1155.
Reformatsky Reaction
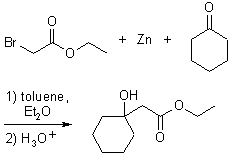
The formation of ester-stabilized organozinc reagents and their addition to carbonyl compounds
Mechanism
Organozinc compounds are prepared from a-halogenesters in the same manner as Grignard Reagents. This reaction is possible due to the stability of esters against organozincs. Due to the very low basicity of zinc enolates, there is hardly any competition from proton transfer, and the scope of carbonyl addition partners is quite broad. In presence of ketones or aldehydes, the organozinc compounds react as the nucleophilic partner in an addition to give ß-hydroxy esters.
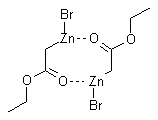
An ester-stabilized organozinc reagent
Recent Literature
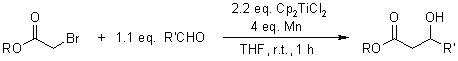
J. D. Parrish, D. R. Shelton, R. D. Little, Org. Lett., 2003, 5, 3615-3617.
Weinreb Ketone Synthesis
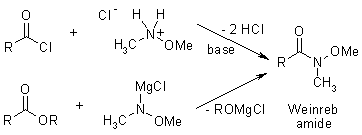
The reaction of esters and carboxylic acid chlorides with organolithium and organomagnesium compounds does not lead to ketones in high yields, because the intermediate ketones are still highly reactive toward the organometallic reagent. However, after derivatisation to the corresponding Weinreb Amide, reaction with organometallics does give the desired ketones, as the initial adduct is stabilized and doesn't undergo further reaction.
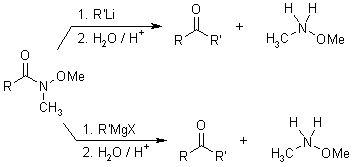
Mechanism
With the usual reaction of organometallic reagents with acid derivatives (ester or acid chloride), the starting materials can add two equivalents of organometallic compound. The ketone generated after the first addition is quite reactive, and there is quite no selectivity between it and the starting acid derivative:
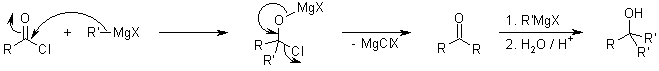
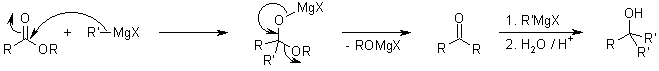
The organometallic adducts of Weinreb Amides are able to form stable chelates, and do not regenerate an electrophilic carbonyl group in situ for further reaction:

Aqueous work up liberates the ketone from this chelate: