This is documented on rhodium but after hours of scanning swim cant find any hive posts on it, can anyone give any notes on it?
Quote
Spent quite a while looking at this and I dont see why it wouldnt work?
Any comments or opinions welcome!
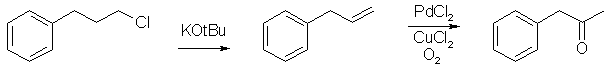
P2P from 3-phenyl-1-chloropropane
Simple dehydrohalogenation to Allylbenzene
And of course Allylbenzene is an oxidation away from our second favourite chemical P2P.
Would love to hear if this is feasable, sounds perfect and OTC if you shop around.