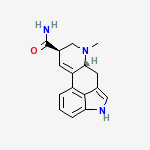
I had taken 4-ho-dmt (psilocin) several times and I found out I don't like it, I don't like the mushrooms trips cause they cause panic, are often times dark and twisted and just plain weird, but i love LSD and mescaline (very much like LSD)...and I love cold water extractions of morning glory with 99% DL tartaric acid added for a ph of 4.0...kicks much ass, but plain water extractions suck and are only sedating compared with the ascorbate/tartrate brews.
Nichols in response to Peter Webster (very important and key):
-------------------------------------------------------------------------------
"Some researchers believe that perhaps all of the content of MG seeds exists as the d-hydroxyethylamide, and it is the extraction/purification/testing procedures which then produce the APPARENT result that the alkaloids are ergine (LSA), isoergine (iso-LSA),and maybe still some hydroxyetylamide."
LSH is a labile adduct of lysergic acid and acetaldehyde = lysergic acid hydroxyethylamide. It is a natural alkaloid found in the seeds, but since it is unstable, it probably comes apart in water solutions unless something is in there to help it keep together. Albert Hofmann discovered lysergic acid hydroxyethylamide in the seeds and shows a color picture of it on page 96 of "LSD, my problem child"...Osmond claims that Hoffman told him that lysergic-acid-hydroxyethylamide is actually "very LSD-like", unlike LSA.
As far as the protection/reducing/anti-oxidant properties of vitamin C: discovered on my own that when 4-aco-dmt is hydrolyzed into 4-ho-dmt, the water solution turns blue indicating the prescense of psilocin which is deteriorating quickly, but when a crushed vitamin C tablet is added, the solution immediately turns back to a clear color...the powerful anti-oxidant and reducing properties of vitamin C or tartaric acid will stop the breakdown of 4-ho-dmt, and that's why it used by those in the know with mushroom tea extractions, to preserve the psilocin and help keep it from breaking down.
Elaboration on why this works in theory I believe:
Acetaldehyde reacts with Lysine to form stable lysine-acetaldehyde adducts at the N-terminal (see photo below) with the reducing agent ascorbate acid present (vitamin c):
http://www.ncbi.nlm.nih.gov/pubmed/3124658
hxxp://www.ncbi.nlm.nih.gov/pubmed/3124658
(there are lots more studies with lysine n-terminal condensing with acetaldehyde to form stable lysine-acetaldehyde adducts...in the prescense of various kinds of reducing agents, even tartaric acid is a powerful reducing agent).
I don't see why the same thing don't happen with LSA and acetaldehyde at the n-terminal with the reducing agent vitamin C as well. the primary difference is that the nitrogen on LSA is an amide, and not a plain old amine, but i've read count-less studies that show that vitamin C and tartaric acid (both powerful reducing agents) act to "stabilize" adducts....so when doing extractions this way....perhaps what we are doing is simply stabilizing the allready present LSH in the seeds so that it re-mains bonded and does not come apart, so that it can be consumed and make it to the brain, as other studies with Lysine and other molecules that bind with acetaldehyde, these studies show that the condensation product with acetaldehyde is made stable with vitamin C and other reducing agents.
What do you guys think? the amino acid lysine on left, LSA on right.
The n-terminal is exactly the same as the n-terminal on LSA.
The acetaldehyde within the seeds condenses with LSA to form lyergic acid hydroxyethylamide, which Hoffman and others found as an active component of the seeds, of course, this adduct is probably "unstable" when it hits alkaline, heated, or maybe even plain water solutions...but I don't see why we can't preserve the adduct in ph=4 acidic ascrobate/tartrate reducing conditions, hell, even add some acetaldehyde in there as well to make damn sure you got plenty of it, lol.
There is ascorbate acid and acetaldehyde in lemons and grapefruit. There is tartaric acid and acetaldehyde in sherry wine etc....Hermes used to get far out trips every time he extracted 8 to 12 g of MG seeds into water with several squirts of lemon juice...the shamans in amazon would use amazonian river water which was allready acidic and ott said that often they would add their MG extraction mixture to "juice". The extractions I did into ph=4 tartaric acid acidified distilled cold water kicked much ass and I still remember those potent trips to this day, but when i used plain water, i got nothing but sedation, it sucked. Fastandbulbous (advanced chem mod) at BL said that when he extacted into water with tartaric acid added, that the trips were potent, and wondered why others didn't use his technique...I discovered this apart from even reading his post on this. morninggloryseed said that when he "ate" the MG and HBWR seeds he got very potent trips, but said that plain water extractions gave him nothing but sedation, Morninggloryseed also mentioned in one of his posts that he had not yet tried to use acidic water, but that it might give different results than his plain water extractions.
When I grew my own MG plants in miracle grow soil, I found the fresh seeds when freshly picked (thousands of seeds per plant) and crushed gave off a very intense fruity aroma, indicating the prescense of much acetaldehyde in the seeds. These seeds resulted in a VERY potent trip when extracted into cold distilled water acidified to ph=4 with DL tartaric acid, they could be kept potent by freezing and vacuum packing. I threw away the seeds when I ran out of room in the freezer but could kick myself now for doing that. I've also found some potent seeds from some on-line suppliers. I did not like the burpee seeds, they sucked. the potency of the seeds can vary by as much as 200% according to morninggloryseed at BL.
the longer the seeds are left crushed and open (exposed to the air) and not put into cold water acidified with either vitamin C or DL tartaric acid (to halt the decomposition) the faster the LSH will decompose into LSA, and acetaldehyde boils off at only 68 degree F (20 degree C)...so this is of utmost importance it seems. Psilocin also decomposes quickly in plain air or water (any where there is oxygen) unless stabilized with vitamin C. Tartaric acid salts are also the preferred method of salting out LSD, keeping it from decomposing in the prescence of oxygen, heat, etc....convert the freebase alkaloids into stable tartrate salt form by extracting the MG seeds into cold water acidified to ph=4 with tartaric acid (use ph paper to measure) as tartaric acid can easily shoot the ph down too low (you don't want it to go much below 4). I found these solutions also kept potent easily for days in the fridge until ready to use.
LSD--------------------------------------CH2CH3---CH2CH3
LAE-32---------------------------------------H------CH2CH3
d-lysergic acid hydroxyethylamide--------H-------CHOHCH3
Notice the extreme similarity of LSH to LAE-32 (in which human experiments were done)
TIHKAL:
----------
LAE-32, N-ethyllysergamide. (D-Lysergic Acid Ethylamide)
Different people have observed and reported different effects, with different routes of
administration. Subcutaneous administrations of from 500 to 750 micrograms have been said to produce a state of apathy and sedation. Clinical studies with dosages of 500 micrograms i.m. were felt to be less effective than the control use of 100 micrograms of LSD. And yet, oral doses of twice this amount, 1.6 milligrams, have been said to produce a short-lived LSD-like effect with none of these negatives.
Wikipedia:
-------------
D-Lysergic Acid Ethylamide, (LAE-32) is a derivative of ergine. It is reported to have some LSD-like effects but is weaker and shorter lasting, with an active dose reported to be between 0.5 and 1.5 milligrams.
1950's animal experiments from Glasser:
----------------------------------------------------
D-Lysergic acid N-(1-hydroxyethyl) amide induces contractions in the isolated uterus of the guinea pig and in the rabbit uterus in situ, showing about 30-50% of the activity of ergometrine. In mice and rabbits it produced the syndrome of central sympathetic stimulation, such as piloerection, mydriasis, and hyperthermia, which suggests that it could have an LSD-like activity, but this hypothesis has not yet been verified by experiments on humans ([60]). He also noted that the animals stood up on their hine legs and touched each other on the noses during the stimulation.
He was going to do human studies, but never got around to it.
Additions to the indole nitrogen (see my old adduct paper) also greatly reduce vasoconstriction (just like the medicine "Sansert") which is just like ergometrine without the severe vasoconstriction, sansert causes profound LSD-like effects in high doses but without the vasoconstriction of ergometrine.
Amino acid Lysine on top, LSA on bottom.
Nichols in response to Peter Webster (very important and key):
-------------------------------------------------------------------------------
"Some researchers believe that perhaps all of the content of MG seeds exists as the d-hydroxyethylamide, and it is the extraction/purification/testing procedures which then produce the APPARENT result that the alkaloids are ergine (LSA), isoergine (iso-LSA),and maybe still some hydroxyetylamide."
LSH is a labile adduct of lysergic acid and acetaldehyde = lysergic acid hydroxyethylamide. It is a natural alkaloid found in the seeds, but since it is unstable, it probably comes apart in water solutions unless something is in there to help it keep together. Albert Hofmann discovered lysergic acid hydroxyethylamide in the seeds and shows a color picture of it on page 96 of "LSD, my problem child"...Osmond claims that Hoffman told him that lysergic-acid-hydroxyethylamide is actually "very LSD-like", unlike LSA.
As far as the protection/reducing/anti-oxidant properties of vitamin C: discovered on my own that when 4-aco-dmt is hydrolyzed into 4-ho-dmt, the water solution turns blue indicating the prescense of psilocin which is deteriorating quickly, but when a crushed vitamin C tablet is added, the solution immediately turns back to a clear color...the powerful anti-oxidant and reducing properties of vitamin C or tartaric acid will stop the breakdown of 4-ho-dmt, and that's why it used by those in the know with mushroom tea extractions, to preserve the psilocin and help keep it from breaking down.
Elaboration on why this works in theory I believe:
Acetaldehyde reacts with Lysine to form stable lysine-acetaldehyde adducts at the N-terminal (see photo below) with the reducing agent ascorbate acid present (vitamin c):
http://www.ncbi.nlm.nih.gov/pubmed/3124658
hxxp://www.ncbi.nlm.nih.gov/pubmed/3124658
(there are lots more studies with lysine n-terminal condensing with acetaldehyde to form stable lysine-acetaldehyde adducts...in the prescense of various kinds of reducing agents, even tartaric acid is a powerful reducing agent).
I don't see why the same thing don't happen with LSA and acetaldehyde at the n-terminal with the reducing agent vitamin C as well. the primary difference is that the nitrogen on LSA is an amide, and not a plain old amine, but i've read count-less studies that show that vitamin C and tartaric acid (both powerful reducing agents) act to "stabilize" adducts....so when doing extractions this way....perhaps what we are doing is simply stabilizing the allready present LSH in the seeds so that it re-mains bonded and does not come apart, so that it can be consumed and make it to the brain, as other studies with Lysine and other molecules that bind with acetaldehyde, these studies show that the condensation product with acetaldehyde is made stable with vitamin C and other reducing agents.
What do you guys think? the amino acid lysine on left, LSA on right.
The n-terminal is exactly the same as the n-terminal on LSA.
The acetaldehyde within the seeds condenses with LSA to form lyergic acid hydroxyethylamide, which Hoffman and others found as an active component of the seeds, of course, this adduct is probably "unstable" when it hits alkaline, heated, or maybe even plain water solutions...but I don't see why we can't preserve the adduct in ph=4 acidic ascrobate/tartrate reducing conditions, hell, even add some acetaldehyde in there as well to make damn sure you got plenty of it, lol.
There is ascorbate acid and acetaldehyde in lemons and grapefruit. There is tartaric acid and acetaldehyde in sherry wine etc....Hermes used to get far out trips every time he extracted 8 to 12 g of MG seeds into water with several squirts of lemon juice...the shamans in amazon would use amazonian river water which was allready acidic and ott said that often they would add their MG extraction mixture to "juice". The extractions I did into ph=4 tartaric acid acidified distilled cold water kicked much ass and I still remember those potent trips to this day, but when i used plain water, i got nothing but sedation, it sucked. Fastandbulbous (advanced chem mod) at BL said that when he extacted into water with tartaric acid added, that the trips were potent, and wondered why others didn't use his technique...I discovered this apart from even reading his post on this. morninggloryseed said that when he "ate" the MG and HBWR seeds he got very potent trips, but said that plain water extractions gave him nothing but sedation, Morninggloryseed also mentioned in one of his posts that he had not yet tried to use acidic water, but that it might give different results than his plain water extractions.
When I grew my own MG plants in miracle grow soil, I found the fresh seeds when freshly picked (thousands of seeds per plant) and crushed gave off a very intense fruity aroma, indicating the prescense of much acetaldehyde in the seeds. These seeds resulted in a VERY potent trip when extracted into cold distilled water acidified to ph=4 with DL tartaric acid, they could be kept potent by freezing and vacuum packing. I threw away the seeds when I ran out of room in the freezer but could kick myself now for doing that. I've also found some potent seeds from some on-line suppliers. I did not like the burpee seeds, they sucked. the potency of the seeds can vary by as much as 200% according to morninggloryseed at BL.
the longer the seeds are left crushed and open (exposed to the air) and not put into cold water acidified with either vitamin C or DL tartaric acid (to halt the decomposition) the faster the LSH will decompose into LSA, and acetaldehyde boils off at only 68 degree F (20 degree C)...so this is of utmost importance it seems. Psilocin also decomposes quickly in plain air or water (any where there is oxygen) unless stabilized with vitamin C. Tartaric acid salts are also the preferred method of salting out LSD, keeping it from decomposing in the prescence of oxygen, heat, etc....convert the freebase alkaloids into stable tartrate salt form by extracting the MG seeds into cold water acidified to ph=4 with tartaric acid (use ph paper to measure) as tartaric acid can easily shoot the ph down too low (you don't want it to go much below 4). I found these solutions also kept potent easily for days in the fridge until ready to use.
LSD--------------------------------------CH2CH3---CH2CH3
LAE-32---------------------------------------H------CH2CH3
d-lysergic acid hydroxyethylamide--------H-------CHOHCH3
Notice the extreme similarity of LSH to LAE-32 (in which human experiments were done)
TIHKAL:
----------
LAE-32, N-ethyllysergamide. (D-Lysergic Acid Ethylamide)
Different people have observed and reported different effects, with different routes of
administration. Subcutaneous administrations of from 500 to 750 micrograms have been said to produce a state of apathy and sedation. Clinical studies with dosages of 500 micrograms i.m. were felt to be less effective than the control use of 100 micrograms of LSD. And yet, oral doses of twice this amount, 1.6 milligrams, have been said to produce a short-lived LSD-like effect with none of these negatives.
Wikipedia:
-------------
D-Lysergic Acid Ethylamide, (LAE-32) is a derivative of ergine. It is reported to have some LSD-like effects but is weaker and shorter lasting, with an active dose reported to be between 0.5 and 1.5 milligrams.
1950's animal experiments from Glasser:
----------------------------------------------------
D-Lysergic acid N-(1-hydroxyethyl) amide induces contractions in the isolated uterus of the guinea pig and in the rabbit uterus in situ, showing about 30-50% of the activity of ergometrine. In mice and rabbits it produced the syndrome of central sympathetic stimulation, such as piloerection, mydriasis, and hyperthermia, which suggests that it could have an LSD-like activity, but this hypothesis has not yet been verified by experiments on humans ([60]). He also noted that the animals stood up on their hine legs and touched each other on the noses during the stimulation.
He was going to do human studies, but never got around to it.
Additions to the indole nitrogen (see my old adduct paper) also greatly reduce vasoconstriction (just like the medicine "Sansert") which is just like ergometrine without the severe vasoconstriction, sansert causes profound LSD-like effects in high doses but without the vasoconstriction of ergometrine.
Amino acid Lysine on top, LSA on bottom.