Extracting different compounds from plants is a great way to obtain those hard to get precursors in viable quantities
Those oz members should be well aware of their good fortunes in this regard with so many potential sources of those useful alkaloids.
In particular is the "Camphorum Cinnamomum" aka the Camphor Laurel,, considered a noxious weed, but so prevalent that large scale extermination is unviable and so it continues to flourish. The oil content of most parts of this plants are high, IMO its got to be one of the oiliest plants around and its this oil which contains one of our favorite catechols...safrole and in very appreciable amounts.
What makes this such a good source is basically its abundance... and its sooo easy to extract. IMO its much easier to 'experiment' with something you have 'enough' of, and/or can get fairly easily so aren't too concerned about spilling a bit
Once you have sourced the plant matter and steam distilled 10kgs (a medium size backpack full) you will have more than enough starting material
Basically you steam distill enough of the root bark to get the get oil
You then fractionate this oil to get your allybenzene (safrole) to be used as seen fit
Here's a fresh take on ghetto steam distilling in the bush
well anywhere you want really, so long as you have water and a heat source to boil it you could do this almost anywhere
A small (40cm) sharpened hand mattock as used to collect bark and small shoots (up to 60cm) which can contain quite a lot of oil. Secateurs tomahawks and othercchopping tools come in handy.
There are no specific yields known as nothing has been weighed prior to distillation so the amounts given in the following procedures are only rough estimates. The oil content of the plant matter varies but guesstimates put the bark as yielding up to 5% oil by weight. A kilo of bark will usually give at least 20gms of oil
With the fractionating yeilds of safrole have varied from around 15-70% of the w/w steam distilled oil.
The reasons behind this are unclear, and may be as varied as things like the time of year the plant was harvested, its location and genetic components. These things aren't easy to discern, some taking years of observation (or intense study) so not really easy to do but not important either. Try out a few different patches and one of them's "gonna work"
Oil has been obtained from all parts of the tree but the berries. Root bark provides the most consistent yields but the young shoots have also given some good amounts of oil. Its clear when you compare and smell different parts of the tree what is 'oilier' The wood itself also can contain a fair amount of oil but its a bitch to chop up enough to get it out But if you have ever taken a chainsaw to one of these trees, you'd know exactly what I mean...if you pick up those shavings you can clearly 'feel' what an oily wood it is.
But if you have ever taken a chainsaw to one of these trees, you'd know exactly what I mean...if you pick up those shavings you can clearly 'feel' what an oily wood it is.
Fortunately, when you find one big camphor you've most likely found a lot more than one and so its easy to mix up what you collect.
The baby plants that have just germinated are high in oil so if (as is common) you come across a few hundred sprouting under a tree they are usually worth picking--again their smell is a good indicator of oil content.
We have tried several variables when sourcing our plants for harvest, and one indicator is a strong smell of camphor in the soil ,around the trunk under the canopy which always seems to indicate high oil contents in the tree/s above. Although its impossible to determine the safrole content the more oil you can get, the better, and if you take your time using a good setup and take your doing the fractional distillation you will get some safrole.
The only things needed, are utensils for chopping up the plant material, a pressure cooker with about 2mts of hose. A receiving container. Something to hold hot things with (rags) Heat and water.
Add the time and place and your good to go
A standard pressure cooker was used, the bark was chopped and roughly ground 'by foot' against a flat stone, into bits as small as reasonably possible; these were about 60% <10mm, 20% <5mm, 20%> 10mm ---
a 'basket' was made with flywire which was more like a very shallow dogs bowl about 3cms above 3-4cms of water, basically to hold the plant matter out of/above the water which seems to work better for this method. Usually
A (in this case, roughly 2mt clear) hose is connected to the pressure release outlet and led into a container (bottle) It can be cooled with wet rags (if necessary i.e. ambient temp is high) but unless its over 30c it usually has condensed by halfway down the tube.
It was noticed that allowing the pressure to build up and caused the bark/plant matter to break down more, so we started cooking for 5-10mins before releasing the steam and this seemed to enhance the extraction, both in speed and yield but it meant more sophistication with collecting the steam...and man that stuff is HOT!
To collect the steam under pressure, a feww modifications were necessary. First, the attatchment of the hose needed to be larger to collect the steam being when releasing pressure.
It's fine just to connect the hose to the steam outlet and just boil away- but we wanted to put the bark under pressure to help break it up and release the oil but we didn't want to wait for it to cool down to attach the 'condenser' hose, but we couldn't attach the hose while the pressure cooker was still capped and so some lateral thought was applied.
First a small wire and string 'jigger' was rigged to open the valve on the pressure cooker with the pull of a string.
Next a 15cm plastic funnel's top circular edge had some tire tube rubber stuck to it as a soft gasket, and then the skinny end of
this funnel was shoved into the end of the hose, fitting quite snugly which is what you want
Then the cooking began and once the pressure had built up (5-10mins) decided by wiggling the pressure release the inverted funnel was placed over the release valve/regulator of the pressure cooker and the 'jiggler' wire activated.
the inverted funnel was placed over the release valve/regulator of the pressure cooker and the 'jiggler' wire activated. 
This was held in place until the pressure had subsided and then the funnel as removed, the hose attached directly to the valve and distillation continued for another 30 mins.
Its often hard to decide when to stop, in our case it a matter of diminishing returns, see
usually if the plant matter is well chopped going over 30-40mins only seems to get you 10-20% more oil (maybe!) with another 30-60mins, whereas recharging the cooker with fresh plant matter will most likely get a similar amount from similar starting material or 100% the same in another 45mins. So if you have lots of stuff to distill this seems to work out.
Be sure to be holding the tube with a thick rag or gloves. IT GETS VERY HOT!
CARE must be taken to avoid inadvertant burns with this pressurized steam..its scary stuff if it runs away on you
Heating was often done on a small fire on location, which was more than adequate and makes it much easier to carry out what you
went for -- go for a picnic with your grrrrlfriend/boyfriend and your favorite pressure cooker,,
Cook up a few bangers while you're at it and no one will be the wiser that you are actually in the process of extracting the oils
for 'making some Love' ;
This extraction gig can be a fun and healthy pastime boys and grrrls, whilst helping you acquire the chemicals required to work on your chemistry skills to be able to synthesize the other chemicals necessary for your well being and development and a balanced state of mind
here's some plants with potential for extraction I discovered quite by accident... for the more eurowasp I guess but interesting nonetheless
Clover
Trifolium repens (Fabaceae) White clover
Trifolium pratense (Fabaceae) Red clover
White and red clover are native to Europe, North Africa and western Asia. They are widely cultivated pasture crops and fodder-plants, valued for their nitrogen fixation by rhizobia, their high protein content, and as one of the main sources of nectar for honey-bees and bumble-bees.
The mild sweet scent of blooming clovers is a charm of the summertime meadows.
Buttery et al. investigated the volatiles from red clover flowers and found that they were dominated by acetophenone (24 %), methyl cinnamate (11 %), 1-phenylethanol (8 %), (E)-beta-ocimene (8 %) and (E)-2-hexenal (6 %) [158].
Etymology: Lat. trifolium, threefold leaf. The clover leaf has several symbolic meanings, it is behind the clubs of playing cards, etc. Lat. repens, creeping; Lat. pratense, growing in the meadows
Privet
Ligustrum vulgare (Oleaceae) European Privet
Ligustrum ovalifolium (Oleaceae) Oval-leaved Privet
PRIVET -> Paa
The genus Ligustrum of the olive family holds around 50 species, most of them from eastern Asia. They are evergreen or semi-evergreen bushes or small trees. Many are used as hedge plants. The only European species is L. vulgare (left photo). Oval-leaved privet from Japan, L. ovalifolium, however, is now more popular for hedging because it keeps its leaves better in winter (right photo).
The creamy-white flowers of European privet are produced in mid-summer and have a characteristic sweetish scent with a sharp nuance - slightly reminiscent of latex (vulcanized) to this author.
Daniel Joulain at Research Labortories, Robertet S.A., investigated the headspace of privet flowers and found that two of the major ingredients are veratrole (14 %) with a creamy, slightly vanilla-like odor, and phenylacetaldehyde (17 %) with a sharp green character. The flowers of oval-leaved privet smell slightly different, phenylacetaldehyde is absent and, in addition to veratrole (4 %), methyl o-anisate (5%) and phloroglucinol trimethyl ether (1 %) are present. Both species also emit linalool (8 % and 5 % respectively) and phenylbutanones, and oval-leaved privet also hotrieneol (0.5 %) [214]
BTW if you have any steam distilling questions I'll be happy to try and answer them...but its a pretty basic task so I'll understand if you dont
f1
Those oz members should be well aware of their good fortunes in this regard with so many potential sources of those useful alkaloids.
In particular is the "Camphorum Cinnamomum" aka the Camphor Laurel,, considered a noxious weed, but so prevalent that large scale extermination is unviable and so it continues to flourish. The oil content of most parts of this plants are high, IMO its got to be one of the oiliest plants around and its this oil which contains one of our favorite catechols...safrole and in very appreciable amounts.
What makes this such a good source is basically its abundance... and its sooo easy to extract. IMO its much easier to 'experiment' with something you have 'enough' of, and/or can get fairly easily so aren't too concerned about spilling a bit

Once you have sourced the plant matter and steam distilled 10kgs (a medium size backpack full) you will have more than enough starting material

Basically you steam distill enough of the root bark to get the get oil
You then fractionate this oil to get your allybenzene (safrole) to be used as seen fit

Here's a fresh take on ghetto steam distilling in the bush

well anywhere you want really, so long as you have water and a heat source to boil it you could do this almost anywhere
A small (40cm) sharpened hand mattock as used to collect bark and small shoots (up to 60cm) which can contain quite a lot of oil. Secateurs tomahawks and othercchopping tools come in handy.
There are no specific yields known as nothing has been weighed prior to distillation so the amounts given in the following procedures are only rough estimates. The oil content of the plant matter varies but guesstimates put the bark as yielding up to 5% oil by weight. A kilo of bark will usually give at least 20gms of oil
With the fractionating yeilds of safrole have varied from around 15-70% of the w/w steam distilled oil.
The reasons behind this are unclear, and may be as varied as things like the time of year the plant was harvested, its location and genetic components. These things aren't easy to discern, some taking years of observation (or intense study) so not really easy to do but not important either. Try out a few different patches and one of them's "gonna work"

Oil has been obtained from all parts of the tree but the berries. Root bark provides the most consistent yields but the young shoots have also given some good amounts of oil. Its clear when you compare and smell different parts of the tree what is 'oilier' The wood itself also can contain a fair amount of oil but its a bitch to chop up enough to get it out
 But if you have ever taken a chainsaw to one of these trees, you'd know exactly what I mean...if you pick up those shavings you can clearly 'feel' what an oily wood it is.
But if you have ever taken a chainsaw to one of these trees, you'd know exactly what I mean...if you pick up those shavings you can clearly 'feel' what an oily wood it is. Fortunately, when you find one big camphor you've most likely found a lot more than one and so its easy to mix up what you collect.
The baby plants that have just germinated are high in oil so if (as is common) you come across a few hundred sprouting under a tree they are usually worth picking--again their smell is a good indicator of oil content.
We have tried several variables when sourcing our plants for harvest, and one indicator is a strong smell of camphor in the soil ,around the trunk under the canopy which always seems to indicate high oil contents in the tree/s above. Although its impossible to determine the safrole content the more oil you can get, the better, and if you take your time using a good setup and take your doing the fractional distillation you will get some safrole.
The only things needed, are utensils for chopping up the plant material, a pressure cooker with about 2mts of hose. A receiving container. Something to hold hot things with (rags) Heat and water.
Add the time and place and your good to go

A standard pressure cooker was used, the bark was chopped and roughly ground 'by foot' against a flat stone, into bits as small as reasonably possible; these were about 60% <10mm, 20% <5mm, 20%> 10mm ---
a 'basket' was made with flywire which was more like a very shallow dogs bowl about 3cms above 3-4cms of water, basically to hold the plant matter out of/above the water which seems to work better for this method. Usually
A (in this case, roughly 2mt clear) hose is connected to the pressure release outlet and led into a container (bottle) It can be cooled with wet rags (if necessary i.e. ambient temp is high) but unless its over 30c it usually has condensed by halfway down the tube.
It was noticed that allowing the pressure to build up and caused the bark/plant matter to break down more, so we started cooking for 5-10mins before releasing the steam and this seemed to enhance the extraction, both in speed and yield but it meant more sophistication with collecting the steam...and man that stuff is HOT!

To collect the steam under pressure, a feww modifications were necessary. First, the attatchment of the hose needed to be larger to collect the steam being when releasing pressure.
It's fine just to connect the hose to the steam outlet and just boil away- but we wanted to put the bark under pressure to help break it up and release the oil but we didn't want to wait for it to cool down to attach the 'condenser' hose, but we couldn't attach the hose while the pressure cooker was still capped and so some lateral thought was applied.
First a small wire and string 'jigger' was rigged to open the valve on the pressure cooker with the pull of a string.
Next a 15cm plastic funnel's top circular edge had some tire tube rubber stuck to it as a soft gasket, and then the skinny end of
this funnel was shoved into the end of the hose, fitting quite snugly which is what you want

Then the cooking began and once the pressure had built up (5-10mins) decided by wiggling the pressure release
 the inverted funnel was placed over the release valve/regulator of the pressure cooker and the 'jiggler' wire activated.
the inverted funnel was placed over the release valve/regulator of the pressure cooker and the 'jiggler' wire activated. 
This was held in place until the pressure had subsided and then the funnel as removed, the hose attached directly to the valve and distillation continued for another 30 mins.
Its often hard to decide when to stop, in our case it a matter of diminishing returns, see
usually if the plant matter is well chopped going over 30-40mins only seems to get you 10-20% more oil (maybe!) with another 30-60mins, whereas recharging the cooker with fresh plant matter will most likely get a similar amount from similar starting material or 100% the same in another 45mins. So if you have lots of stuff to distill this seems to work out.

Be sure to be holding the tube with a thick rag or gloves. IT GETS VERY HOT!
CARE must be taken to avoid inadvertant burns with this pressurized steam..its scary stuff if it runs away on you

Heating was often done on a small fire on location, which was more than adequate and makes it much easier to carry out what you
went for -- go for a picnic with your grrrrlfriend/boyfriend and your favorite pressure cooker,,
Cook up a few bangers while you're at it and no one will be the wiser that you are actually in the process of extracting the oils
for 'making some Love' ;
This extraction gig can be a fun and healthy pastime boys and grrrls, whilst helping you acquire the chemicals required to work on your chemistry skills to be able to synthesize the other chemicals necessary for your well being and development and a balanced state of mind

here's some plants with potential for extraction I discovered quite by accident... for the more eurowasp I guess but interesting nonetheless

Clover
Trifolium repens (Fabaceae) White clover
Trifolium pratense (Fabaceae) Red clover
White and red clover are native to Europe, North Africa and western Asia. They are widely cultivated pasture crops and fodder-plants, valued for their nitrogen fixation by rhizobia, their high protein content, and as one of the main sources of nectar for honey-bees and bumble-bees.
The mild sweet scent of blooming clovers is a charm of the summertime meadows.
Buttery et al. investigated the volatiles from red clover flowers and found that they were dominated by acetophenone (24 %), methyl cinnamate (11 %), 1-phenylethanol (8 %), (E)-beta-ocimene (8 %) and (E)-2-hexenal (6 %) [158].
Etymology: Lat. trifolium, threefold leaf. The clover leaf has several symbolic meanings, it is behind the clubs of playing cards, etc. Lat. repens, creeping; Lat. pratense, growing in the meadows
Privet
Ligustrum vulgare (Oleaceae) European Privet
Ligustrum ovalifolium (Oleaceae) Oval-leaved Privet
PRIVET -> Paa
The genus Ligustrum of the olive family holds around 50 species, most of them from eastern Asia. They are evergreen or semi-evergreen bushes or small trees. Many are used as hedge plants. The only European species is L. vulgare (left photo). Oval-leaved privet from Japan, L. ovalifolium, however, is now more popular for hedging because it keeps its leaves better in winter (right photo).
The creamy-white flowers of European privet are produced in mid-summer and have a characteristic sweetish scent with a sharp nuance - slightly reminiscent of latex (vulcanized) to this author.
Daniel Joulain at Research Labortories, Robertet S.A., investigated the headspace of privet flowers and found that two of the major ingredients are veratrole (14 %) with a creamy, slightly vanilla-like odor, and phenylacetaldehyde (17 %) with a sharp green character. The flowers of oval-leaved privet smell slightly different, phenylacetaldehyde is absent and, in addition to veratrole (4 %), methyl o-anisate (5%) and phloroglucinol trimethyl ether (1 %) are present. Both species also emit linalool (8 % and 5 % respectively) and phenylbutanones, and oval-leaved privet also hotrieneol (0.5 %) [214]
BTW if you have any steam distilling questions I'll be happy to try and answer them...but its a pretty basic task so I'll understand if you dont

f1







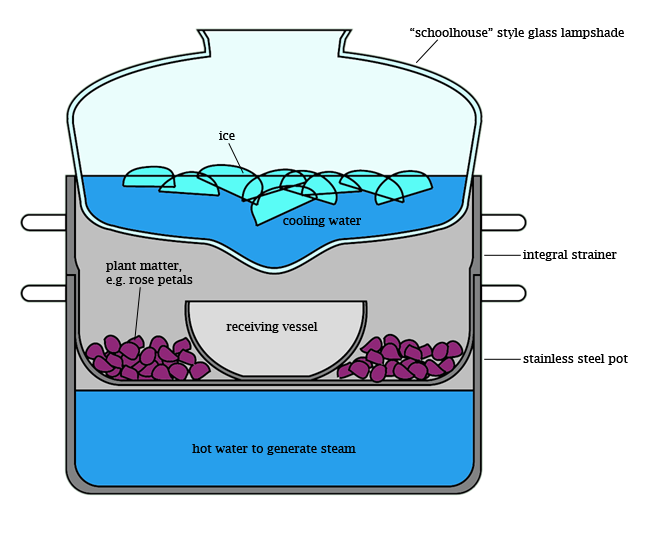

 and the flywire always seemed to be around in the car
and the flywire always seemed to be around in the car  so ghetto it was
so ghetto it was 


