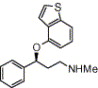
This is just something I have been playing around with in my head for a while. I am curious as to what research has been done on phenethylamines containing hetrocyclic aromatic backbones. A number of active analogs have been created by altering the 5 membered methylenedioxy ring in MDA. These analogs such as 6-APB[1] and 5-API[2] have 5 membered rings containing both oxygen and nitrogen atoms, forming furan and pyrrole rings respectively. Both of these modifications have resulted in a similar or significant increase in potency.
My question is have any sulfur containing analogs of MDA been synthesised and if so have they any activity in humans? Shulgin et. al has synthesised a range of thiophenethylamines (2C-T's and ALEPH's) which have some unique characteristic. They also exhibit a much increased activity. For example 2C-O-4 (http://isomerdesign.com/PiHKAL/read.php?domain=pk&id=35) is inactive where as the thio analog is active at <10mg. From what I can see David E. Nichols theorizes that thiophenethylamines should be more active as they are easily subjected to oxidative metabolism.
An interesting first choice would be the 3,4-methylenedithioamphetamine where both oxygen atoms in MDA are substituted with sulfur atoms. benzene-1,2-dithiol (17534-15-5) appears to be commercial available. Pardon my naivety but I see know reason the desired 3,4-methylenedithiole could not be synthesised from this starting material analogously to the synthesis of MDA from catechol.
Proposed Synthesis:
I have no references for the methylation of thiols ATM, but I don't see why I can't be methylated with dibromomethane in the presence of a PTC as is possible with substituted catechol's[3]. Please correct me if I am completely wrong. Either way it is possible to form the 1,3-benzodithiol by some means. Once benzodithiol has been obtained it should be possible to obtain the respective phenylpropan-2-one by Friedel-Crafts alkylation with 2-Nitropropene.[4] Finally this P2P can be subjected to reductive amination by the known means to the desired amine. It should be possible to obtain 3,4-methyleneoxathioamphetamine by just starting with the 2-hydroxythiophenol instead and methylating to 1,3-benzoxothiole?? (Unsure of correct IUPAC name).
I'm not sure why more research hasn't been done into these sulfur based analogs considering the potency and effect of the 2C-T's. These substances should also be legal, at least under the UK Misuse of Drugs Act. Please let me know what you think of this. Do you think these would be active and if so do they look worthwhile synthesizing?
3,4-methylenedithioamphetamine (MDTA?)

3,4-methyleneoxythioamphetamine

6-(2-aminopropyl)benzothiophene

Edit: It appears that 1,3-benzothiophene-5-carbaldehyde (CAS#: 10133-30-9) is commercially available. This should make this analog very easy to synthesize. Simply condense with nitroethane and then reduce as standard to the amine.
References:
[1] 6-APB - http://en.wikipedia.org/wiki/6-APB
[2] 5-API - http://en.wikipedia.org/wiki/5-%282-Aminopropyl%29indole
[3] PTC Methylenation of Catechol (Using Adogen 464) - http://www.erowid.org/archive/rhodium/chemistry/methylenation.html#PTC1
[4] Synthesis of MDP2P From 1,3-Benzodioxole by Friedel-Crafts Alkylation with 2-Nitropropene - http://www.erowid.org/archive/rhodium/chemistry/mdp2p.nitroalkylation.html
My question is have any sulfur containing analogs of MDA been synthesised and if so have they any activity in humans? Shulgin et. al has synthesised a range of thiophenethylamines (2C-T's and ALEPH's) which have some unique characteristic. They also exhibit a much increased activity. For example 2C-O-4 (http://isomerdesign.com/PiHKAL/read.php?domain=pk&id=35) is inactive where as the thio analog is active at <10mg. From what I can see David E. Nichols theorizes that thiophenethylamines should be more active as they are easily subjected to oxidative metabolism.
Quote
The ability of a compound to produce hallucinatory disturbances may be lost when the 4-substituent is resistant to oxidation. The concept that facile oxidation of the 4-function might be involved in the production of hallucinations by psychotomimetic amphetamine derivatives may receive support through study of an amphetamine derivative with a 4-substituent that is unusually susceptible to oxidative metabolism.
The sulfur atom would appear to be an excellent choice for such a substituent. Metabolically, it should retain the chemical properties of oxygen in reactions such as S-demethylation but should allow the formation of sulfoxides and sulfones as products of oxidative metabolism12. The lipophilicity, electronic character, and size of sulfur also seem ideal.
David E. Nichols & Alexander T. Shulgin - Sulfur Analogs of Psychotomimetic Amines, J. Pharm. Sci. 65(10), 1554-1555 (1976)
An interesting first choice would be the 3,4-methylenedithioamphetamine where both oxygen atoms in MDA are substituted with sulfur atoms. benzene-1,2-dithiol (17534-15-5) appears to be commercial available. Pardon my naivety but I see know reason the desired 3,4-methylenedithiole could not be synthesised from this starting material analogously to the synthesis of MDA from catechol.
Proposed Synthesis:
I have no references for the methylation of thiols ATM, but I don't see why I can't be methylated with dibromomethane in the presence of a PTC as is possible with substituted catechol's[3]. Please correct me if I am completely wrong. Either way it is possible to form the 1,3-benzodithiol by some means. Once benzodithiol has been obtained it should be possible to obtain the respective phenylpropan-2-one by Friedel-Crafts alkylation with 2-Nitropropene.[4] Finally this P2P can be subjected to reductive amination by the known means to the desired amine. It should be possible to obtain 3,4-methyleneoxathioamphetamine by just starting with the 2-hydroxythiophenol instead and methylating to 1,3-benzoxothiole?? (Unsure of correct IUPAC name).
I'm not sure why more research hasn't been done into these sulfur based analogs considering the potency and effect of the 2C-T's. These substances should also be legal, at least under the UK Misuse of Drugs Act. Please let me know what you think of this. Do you think these would be active and if so do they look worthwhile synthesizing?
3,4-methylenedithioamphetamine (MDTA?)

3,4-methyleneoxythioamphetamine

6-(2-aminopropyl)benzothiophene

Edit: It appears that 1,3-benzothiophene-5-carbaldehyde (CAS#: 10133-30-9) is commercially available. This should make this analog very easy to synthesize. Simply condense with nitroethane and then reduce as standard to the amine.
References:
[1] 6-APB - http://en.wikipedia.org/wiki/6-APB
[2] 5-API - http://en.wikipedia.org/wiki/5-%282-Aminopropyl%29indole
[3] PTC Methylenation of Catechol (Using Adogen 464) - http://www.erowid.org/archive/rhodium/chemistry/methylenation.html#PTC1
[4] Synthesis of MDP2P From 1,3-Benzodioxole by Friedel-Crafts Alkylation with 2-Nitropropene - http://www.erowid.org/archive/rhodium/chemistry/mdp2p.nitroalkylation.html