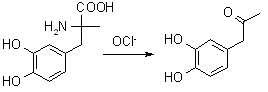"This thread has gone way off topic, though, and the second half probably needs split off and moved to Drug Synthesis?"
LOL, yes it has been a big weekend. Regarding comments about the complexity of this route, I think when you see how straight forward it is in practice you will be suprised. Making a sodium nitrite and hydrochloric acid solution, then stirring that with phenylalanine after a couple of minutes you work up phenyllactic acid (quantitative yield). The phenyllactic acid is stirred with a solution of hydroxylamine and sodium hydroxide overnight(quantitative yield), then neutralized and sulfuric acid added to catalyze the Beckamnn rearrangement then the amide is worked up(anywhere from 75% to quantitative yield). The amide is stirred with hypochlorite and sodium hydroxide for half an hour and worked up(anywhere from 20% to 90% yield, unknown until trial runs are undertaken), hydrobromic acid or hydroiodic acid is added to the amine and stirred(in excess of 75% yield). A Lithium battery is opened up and the Lithium pilfered, all one has to do is get or make some methyl iodide and add the bromo-amine and lithium in solvent and the alkylhalide. Amphetamine is worked up after the reaction!
Sodium Nitrite (easy)
Hydrochloric acid (easy)
Phenylalanine (D or L or both) (easy)
Sodium Hydroxide (easy)
Sulfuric acid (easy)
Sodium Hypochlorite [chlorox] (easy)
Hydroxylamine (medium)
Hydrogen halide (medium to hard)
Methyl Iodide (hard)
Considering peeps used to rob a Buffalo to convert a couple of packets of cold pills, the methyl iodide does not seem that bad. Red P and iodine or the three hard to get things above?
"I have been considering a similar route to MDMA via L-DOPA. Apparently psychokitty had been working on a similar path from methyldopa which would avoid the grignard step, but methyldopa is restricted in my country."
LOL, methyldopa would be priceless to get a hold of, and everyone (including the LE) knows it!!

Look at that! A simple Strecker degradation with hypochlorite would produce 3,4-dihydroxyphenyl-2-proanone!!!
There would be no need for the Grignard as there is already an alpha methyl group present in Methyldopa.
As for the Phenylalanine->Phenylacetaldehyde, I have been discussing that route for some time now. And there has always been inherent problems with that procedure that would need some working (polymerization etc). Although more steps in the route above it is more user friendly.
LOL, yes it has been a big weekend. Regarding comments about the complexity of this route, I think when you see how straight forward it is in practice you will be suprised. Making a sodium nitrite and hydrochloric acid solution, then stirring that with phenylalanine after a couple of minutes you work up phenyllactic acid (quantitative yield). The phenyllactic acid is stirred with a solution of hydroxylamine and sodium hydroxide overnight(quantitative yield), then neutralized and sulfuric acid added to catalyze the Beckamnn rearrangement then the amide is worked up(anywhere from 75% to quantitative yield). The amide is stirred with hypochlorite and sodium hydroxide for half an hour and worked up(anywhere from 20% to 90% yield, unknown until trial runs are undertaken), hydrobromic acid or hydroiodic acid is added to the amine and stirred(in excess of 75% yield). A Lithium battery is opened up and the Lithium pilfered, all one has to do is get or make some methyl iodide and add the bromo-amine and lithium in solvent and the alkylhalide. Amphetamine is worked up after the reaction!
Sodium Nitrite (easy)
Hydrochloric acid (easy)
Phenylalanine (D or L or both) (easy)
Sodium Hydroxide (easy)
Sulfuric acid (easy)
Sodium Hypochlorite [chlorox] (easy)
Hydroxylamine (medium)
Hydrogen halide (medium to hard)
Methyl Iodide (hard)
Considering peeps used to rob a Buffalo to convert a couple of packets of cold pills, the methyl iodide does not seem that bad. Red P and iodine or the three hard to get things above?
"I have been considering a similar route to MDMA via L-DOPA. Apparently psychokitty had been working on a similar path from methyldopa which would avoid the grignard step, but methyldopa is restricted in my country."
LOL, methyldopa would be priceless to get a hold of, and everyone (including the LE) knows it!!

Look at that! A simple Strecker degradation with hypochlorite would produce 3,4-dihydroxyphenyl-2-proanone!!!
There would be no need for the Grignard as there is already an alpha methyl group present in Methyldopa.
As for the Phenylalanine->Phenylacetaldehyde, I have been discussing that route for some time now. And there has always been inherent problems with that procedure that would need some working (polymerization etc). Although more steps in the route above it is more user friendly.