Bromo-DragonFLY
IUPAC name; (1-(8-Bromobenzo[1,2-b;4,5-b]difuran-4-yl)-2-aminopropane
Other names; Bromo-benzodifuranyl-isopropylamine, Bromo-DragonFLY
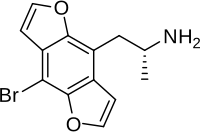
Molecular formula; C13-H12-Br-N-O2
Molecular mass; 294.14g/mol
Melting point; hydrochloride decomposes at 240*C
Drugs Acting on 5-Hydroxytryptamine Receptors
Dihydrobenzofuran Analogues of Hallucinogens. 3.1 Models of 4-Substituted (2,5-Dimethoxyphenyl)alkylamine Derivatives with Rigidified Methoxy Groups2
Aaron P. Monte,‡ Danuta Marona-Lewicka, Matthew A. Parker, David B. Wainscott,† David L. Nelson,† and
David E. Nichols
J. Med. Chem. 1996, 39, 2953-2961
Download paper
A novel (benzodifuranyl)aminoalkane with extremely potent activity at the 5-HT2A receptor.
Parker, M.A.; Marona-Lewicka, D.,; Lucaites, V.L.; Nelson, D.L.; Nichols, D.E.;
J Med Chem 1998, 41, 26, 5148
Download Paper

Alkylation of hydroquinone (I) with 1-bromo-2-chloroethane and K2CO3 in acetone gave the bis(2-chloroethyl)ether (II), which was then brominated in the presence of Fe in CCl4 to provide dibromocompound (III). Lithiation, followed by intramolecular cyclization, upon treatment with two equivalents of n-BuLi in THF at 0 C furnished the tetrahydrobenzodifuran (IV). Subsequent formylation with dichloromethyl methyl ether in the presence of SnCl4 yielded aldehyde (V). This was condensed with nitroethane in the presence of ammonium acetate, and the resulting nitropropene compound (VI) was reduced with LiAlH4 to afford the aminopropane (VII). Protection of (VII) with trifluoroacetic anhydride and Et3N gave trifluoroacetamide (VIII), and then bromination in AcOH afforded bromide (IX). The aromatic benzodifuran (X) was obtained by dihydrogenation with dichlorodicyanobenzoquinone (DDQ) in toluene, and finally, the amide was deprotected by hydrolysis with NaOH to afford the target amine, which was isolated as the hydrochloride salt.
Phenmetrazine
IUPAC name; 3-methyl-2-phenylmorpholine
Other names; Oxazimedrine, Preludin(US), Anorex(US), Cafilon(Jpn), Marsin(Isr)
(V)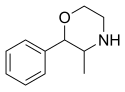
Molecular formula; C11-H15-N-O
Molecular mass; 177.2456 g/mol
Drugs Acting as Norepinephrine and Dopamine releasing agents
Process for the production of substituted Morpholines
US Patent 2,835,669
Download patent
Amine alkylation between a-bromopropiophenone(I) and N-benzylmonoethanolamine(II) produces 2-(benzyl(2-hydroxyethyl)amino)-1-phenylpropan-1-one(III). Hydrogenation in the presence of palladium on charcoal affords deprotection and reduction giving 2-(2-hydroxyethylamino)-1-phenylpropan-1-ol (IV), upon standing with conc acid (H2SO4 or HCl) overnight (or warming for 6 hours) ring closure leading to the required morpholine derivative (3-methyl-2-phenylmorpholine (V)) is complete.
(I) (II)
(II)
(III) (IV)
(IV)
These are a couple of syntheses that are a bit more difficult to come across, I am currently researching the 3,4-methylenedioxy analog of Phenmetrazine if my research keeps going well I hope to produce it soon.
IUPAC name; (1-(8-Bromobenzo[1,2-b;4,5-b]difuran-4-yl)-2-aminopropane
Other names; Bromo-benzodifuranyl-isopropylamine, Bromo-DragonFLY

Molecular formula; C13-H12-Br-N-O2
Molecular mass; 294.14g/mol
Melting point; hydrochloride decomposes at 240*C
Drugs Acting on 5-Hydroxytryptamine Receptors
Dihydrobenzofuran Analogues of Hallucinogens. 3.1 Models of 4-Substituted (2,5-Dimethoxyphenyl)alkylamine Derivatives with Rigidified Methoxy Groups2
Aaron P. Monte,‡ Danuta Marona-Lewicka, Matthew A. Parker, David B. Wainscott,† David L. Nelson,† and
David E. Nichols
J. Med. Chem. 1996, 39, 2953-2961
Download paper
A novel (benzodifuranyl)aminoalkane with extremely potent activity at the 5-HT2A receptor.
Parker, M.A.; Marona-Lewicka, D.,; Lucaites, V.L.; Nelson, D.L.; Nichols, D.E.;
J Med Chem 1998, 41, 26, 5148
Download Paper

Alkylation of hydroquinone (I) with 1-bromo-2-chloroethane and K2CO3 in acetone gave the bis(2-chloroethyl)ether (II), which was then brominated in the presence of Fe in CCl4 to provide dibromocompound (III). Lithiation, followed by intramolecular cyclization, upon treatment with two equivalents of n-BuLi in THF at 0 C furnished the tetrahydrobenzodifuran (IV). Subsequent formylation with dichloromethyl methyl ether in the presence of SnCl4 yielded aldehyde (V). This was condensed with nitroethane in the presence of ammonium acetate, and the resulting nitropropene compound (VI) was reduced with LiAlH4 to afford the aminopropane (VII). Protection of (VII) with trifluoroacetic anhydride and Et3N gave trifluoroacetamide (VIII), and then bromination in AcOH afforded bromide (IX). The aromatic benzodifuran (X) was obtained by dihydrogenation with dichlorodicyanobenzoquinone (DDQ) in toluene, and finally, the amide was deprotected by hydrolysis with NaOH to afford the target amine, which was isolated as the hydrochloride salt.
Phenmetrazine
IUPAC name; 3-methyl-2-phenylmorpholine
Other names; Oxazimedrine, Preludin(US), Anorex(US), Cafilon(Jpn), Marsin(Isr)
(V)

Molecular formula; C11-H15-N-O
Molecular mass; 177.2456 g/mol
Drugs Acting as Norepinephrine and Dopamine releasing agents
Process for the production of substituted Morpholines
US Patent 2,835,669
Download patent
Amine alkylation between a-bromopropiophenone(I) and N-benzylmonoethanolamine(II) produces 2-(benzyl(2-hydroxyethyl)amino)-1-phenylpropan-1-one(III). Hydrogenation in the presence of palladium on charcoal affords deprotection and reduction giving 2-(2-hydroxyethylamino)-1-phenylpropan-1-ol (IV), upon standing with conc acid (H2SO4 or HCl) overnight (or warming for 6 hours) ring closure leading to the required morpholine derivative (3-methyl-2-phenylmorpholine (V)) is complete.
(I)
 (II)
(II)
(III)
 (IV)
(IV)
These are a couple of syntheses that are a bit more difficult to come across, I am currently researching the 3,4-methylenedioxy analog of Phenmetrazine if my research keeps going well I hope to produce it soon.










 .
.