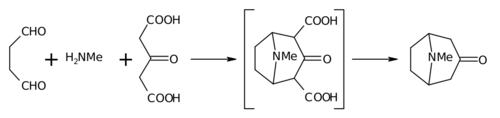
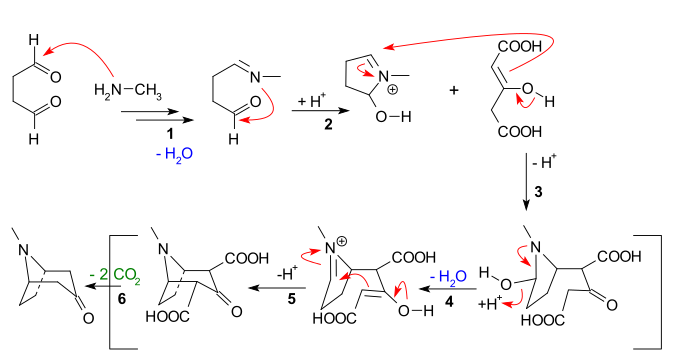
That was not organic reaction vol 1..... But the paper I posted on the first page, and my comments! The Mannich reaction is covered in Organic reactions and even piperidone synthesis from the mannich reaction covered but not that method!
The Preparation of Some Piperidine Derivatives by the Mannich ReactionBY C. R. NOLLERAN D V. BALIAH
A number of piperidones have been prepared by
the Mannich reaction .It should be possible to
extend the use of this reaction for the synthesis of
a large variety of 4-piperidones with different substituents
in the 1,2,3,5 and 6 positions.
Accordingly
glacial acetic acid was tried as a solvent
and reaction took place rapidly, the isolation of
pure products was easy, and the yields were very
satisfactory.
http://127.0.0.1/Naf1/4-piperidones-one-pot-1.pdf------------------------------------------------------------------------------------
Organic Reactions vol 1
"Aldehydes other than formaldehyde may be used in certain condensations
of the Mannich type. Those which have been studied are
acetaldehyde, phenylacetaldehyde, benzaldehyde, and anisaldehyde.
These have been employed successfully with acetone, cyclohexanone,
and esters of acetonedicarboxylic acid. The reactions appear to be
limited to ammonia and primary amines and their salts. With acetone,
aniline, and benzaldehyde a piperidone is obtained.4"*
An open chain product is obtained from cyclohexanone, phenylacetaldehyde
and benzylamine
Substituted piperidones are always produced when esters of acetonedicarboxylic
acid are employed, as in the reaction of the methyl ester
with allylamine and benzaldehyde.
Similar piperidones have been obtained by substituting for allylamine
the following: ammonia,4" methylamine,34 ethylamine,4d and /3-hydroxyethylamine;
84 by employing acetaldehyde, instead of benzaldehyde,
with ammonium bromide,39 methylamine,39 benzylamine,39 and /3-phenylethylamine;
39 and by using allylamine, anisaldehyde, and methyl
acetonedicarboxylate .34
http://asterix.msp.univie.ac.at/.Chemistry%20eBooks%20Collection/Organic/Organic%20reactions%20-%20Vol%201%20-%20Adams.pdf-------------------------------------------------------------------------------------------------------------------
"sure the yeilds would be shit but when your dealing with oh-me-fentanyl low yeilds don't really matter"
Exactly!