Herein you shall find a guide to good practise while hunting and IDing mushrooms.
The intent being to have fun, have a good meal/trip and not to shit your liver out in liquid form.
When hunting, harvest by digging up the mushroom using a knife or spoon, do not yank them up by the stem, or rip the caps alone off, the stem, and base of the stem are vital diagnostic characteristics for telling what you have. Is a volva (cup-like structure) present at the base of the stem? this is diagnostic of two families, Amanita, and Volvariella, which are unrelated (confusion of the edible straw mushroom popular with orientals, and often used in chinese food, Volvariella spp. with deathcaps of varying species such as Amanita phalloides, A.ocreata and some of the similar looking Amanita subsection Phalloidea members is a major cause of death amongst eastasian immigrant mushroom hunters coming to the states or europe who make a dangerous fuckup as they look quite similar)
Be very wary and careful of any mushroom with a volva, do not bag said mushrooms in the same container you are taking any harvest that will be eaten home in unless it is to be fed to an enemy.
Is a ring present on the stem? in some species this tends to disintegrate or fray with age, and in some others, it is moveable up and down the stem if you do so carefully, IIRC, the parasol mushroom, Lepiota procera, a large member of the Lepiota family, which unlike some,which are deadly poisonous, is edible, and indeed, one I rather like to eat, can have its ring slid/rolled up and down the stem if you do it carefully. A few fungi have double rings, but this isn't particularly common.
Pay close attention to the morphology of the cap, and stem. Does the cap have a peelable surface, if the species has a cap. Some do not. Some species one can peel it off, such as the slippery jack bolete (suillus luteus), the related larch bolete (also in the genus Suillus), as do the death cap (Amanita phalloides) and common button mushrooms bought in stores, of the genus Agaricus, most commonly A.bitorquis (ick!....I don't like store bought mushrooms at all, apart from shiitake, oysters etc.) Whilst others do not, for example Mycena spp. and a good few of the Coprinus species. Does the cap become viscid when wet? or is it permanently sticky/glutinous, or is it not so at all? Note texture and smell of the fruitbody, it can help to crush some to release any smell more.
Some fungi have very diagnostic odours, such as in certain Russulas, the smell of decaying shellfish for R.foetens, in R.fellea, there is a pronounced smell of geranium, or in the case of, for example, Russula queletii, the scent of fresh apples.
Check gill structure (gills decurrent, running down the stem? adnexed? adnate? detatched from the stem? texture and resilience of gills, are they waxy? firm? do they break easily? pores like a bolete? some fungi, including a few absolutely excellent edibles, the hedgehog fungi, have pointy spines pointing downwards from the underside of the cap) Some fungi have no true gills, but instead, raised, forking, ridge-like pseudogills forming the fertile area, and some, have no gills at all, such as the horn of plenty/black funnel of death, known to be a very sought after edible, meant to be particularly good in soup or stew, after being dried for storage like one can with porcini/cep/Boletus edulis, but I have never been lucky enough to find any
In some genera, SOME, mind you, but not all, taste is important. Russula, Lactarius, and to a lesser extent, Boletus and their relatives such as Suillus and Chalciporus (C.piperatus for example, the peppery bolete, I use as a spice, dried and ground, just like I use chillis or pepper, its hot and firey, but in a different way to either spice)
The procedure for tasting is to break off a small piece of cap, and nibble it between the teeth for a short time, make sure it gets on the tongue, is it sweet? acrid? or is there a specific taste of something? In particular with Russula and Lactarius species, this test is borderline vital, and in these two families, there are none that a mere taste will kill. Although actually eating a scant handful of Lactarius species has done so in the past, and Russula subnigricans, the blackening russula (not to be confused with R.nigricans or R.nigrescens) contains a potent myotoxin, cycloprop-2-ene-1-carboxylic acid, causing severe rhabdomyolysis and subsequent kidney failure, this species can be lethal, and most painfully, slowly so at that.
Taste, but do not swallow. Spit.
Some fungi take time for a taste to develop, others start one way, then turn another, such as starting mild, but with a rapid, or slow development of an intense acrid burning sensation in the case of some of the Lactarius (milk caps).
In the case of this family, milk cap, comes from their bleeding a milky liquid substance when injured. Cause this to happen if you think you may have one from this genus, observe the consistency and color of the 'milk', some change color on exposure to air after a few minutes, blotting some milk onto white, absorbent paper such as a tissue, or bit of blotting paper can aid the development of the change. There are some good edibles to be had here, one or two species, such as L.torminosus, the wooly milk-cap may be fatal if eaten, but most likely will just cause severe GI irritation and accompanying miserable symptoms for a day or two. The toxins are most likely in most cases to be sesquiterpene compounds, as are those in many of the acrid Russulas (brittlegills). The Russulas, are, with the exception of the myotoxin-bearing R.subnigricans, not fatal, barring some freak ill luck, and besides, are so acrid in many cases for the toxic ones (in fact almost all toxic russilas, bar that species have a hot, burning taste. With very few exceptions, all burning russulas are for the chuck, and some consider it a general rule that all mild ones are edible, I do not rely on rules of thumb though to be exact, not when it comes to potential toxicity, but its very unlikely that you will be poisoned by a mild-tasting Russula.
There are some genera that you simply do not taste. Amanitas, don't do it. Inocybes, don't do it-there aren't any worth eating, and quite possibly none safe to eat either. The entire genus is chock-full of muscarine, with a few exceptions. Most are incredibly hard to ID, and impossible to do so without a microscope to check the spores, gill microstructure and hyphae out. Fun to pick for the challenge of an ID, but certainly not for eating. One mycologist became so ill after tasting a tiny piece of Amanita xanthocephala, an unusual species, not amatoxic, but most virulent nevertheless, that he broke his toes trying to get to the bathroom he was in such a hurry. And tasting a small piece of what turned out to be Podostroma cornu-damae, an oriental club fungus/coral type bugger would at the least burn and blister like mustard gas, and quite possibly prove fatal, although fungi of such extreme toxicity are the exception rather than the rule, and tasting even an amatoxic species by accident is very unlikely to kill you, although may nevertheless make you severely ill and cause liver damage to some degree.
Eat no mushroom that displays bioluminescence. All of them I am aware of that do phosphoresce in the dark, such as Lampteromyces japonicus, and Omphalotus species (the jack'o'lantern fungi) are poisonous. Interestingly the latter family produce their phosphorescence in the same way that fireflies do, the reaction between luciferin and luciferase. Eat something that glows in the dark, and you will regret it. Won't kill you, but will cause very severe GI upset.
Don't taste any Lepiota species either, many contain the same deadly amatoxins that the Amanita (subsection Phalloidea) do. Likewise for Galerina, and Conocybe, there are psilocybin-bearing members of both, but likewise amatoxin carrying ones, avoid tasting or eating these families in their entirety, its not worth repeatedly squirting your liver and kidneys down the inside of your trouser leg in a cloud of blood and cholera-esque chunky fecal soup until you shrivel up, turn yellow, and die over several weeks.
More on toxicity later, I'll give a breakdown of the main groups of toxic compounds, and the species (not totally comprehensive, its a bit too much to expect of me to know and to remember to name every single species in every family containing a given toxin or toxins, but I will give a wide range, of both the common ones, and a fair few more unusual and little nown nasties, where they are found and how they work)
Take note also of the habitat-is it growing on wood, if so, if you can tell, what kind of wood? growing on turds? or near, but not ON shit. In lawns/grassland? woodland? many fungi are what is called mycorrhizal, meaning they form an intimate composite structure between plant roots and the mushroom mycelium. If growing in association with plants, take samples of the nearby (for a few meters) trees and shrubs, most commonly a mycorrhizal fungus will form an association between a tree or woody shrub, macrofungi growing in association with herbs are much less common, and in the case of ergots, take at least one entire grass plant, leaving sclerotia in the seedhead for ID, as the host species often defines the parasite, C.purpurea excepted, which is unusual amongst the Clavicipitacea in being a bit of a whore really, where as others such as C.fusiformis are more picky, there is even one species specialising in bamboo.
Take spore prints, this is a very important step if you are uncertain of the identification. A microscope is helpful too, for some genera, it is vital if you are to ID down to species level, such as the genus Inocybe in particular (not that any of these are ones you want to eat, although a few contain psilocybin and psilocin, I.aeruginescens contains a novel tryptamine, the trimethyltryptammonium quat of psilocybin, although this will not pass the BBB, and is a POTENT 5HT6 agonist, which will make you feel very, very, very severely nauseated and cause quite spectacular vomiting, if its active in vivo at all, compare with ondansetron etc. the antinauseant drugs. The tryptamine bearing Inocybes in many cases contain very low concentrations of actives, avoid them IMO, as most species are full of the cholinergic neurotoxin muscarine, in many cases in large quantities, in some, enough to prove fatal) Pretty much all Inocybes are small to moderate sized LBMs (little brown mushrooms) with fibrous cap surfaces and very hard to ID.
Note spore color, and miscroscopic characteristics if you have a microscope. I need a new one :/
Next-Colorimetric reactions and reagent testing.
Testing fungi with chemicals is a very useful diagnostic tool, this can be done in the field in many cases.
First and foremost-the Meixner test. This isn't a test for species ID, but rather, for the presence of a specific group of toxins, the deadly cytotoxic amatoxins, on which, more information later on in my guide. It is sensitive to 2mcg of amatoxin (calc. as amatoxin-a, alpha-amanitin), a typical Amanita phalloides fruitbody of medium size contains between 4-7mg of total amatoxins, maybe a little more, maybe a little less, fungi are very variable as to chemotype, A.ocreata may contain even more. 10mg/fruitbody-1mg-gram wet tissue on average, or slightly more. 0.1mg/kg is more than enough to kill.
Take a small sample of mushroom cap, or if somebody has eaten the mushroom and an amatoxic species is suspected or even wondered about but only bits of stem or other waste is available, use that, and a slip of paper, this must be high in lignin, or the test will fail to work, or at best will work very poorly, indeed this has failed before, with the result of the direst consequences for the poor bastard the test had been done on behalf of when lab filter paper was used. Newspaper is an excellent type to use.
Mash the sample up good onto the paper, after drawing a pencil circle to tell you where the spot is going to appear if the test is positive, around the area. Work the sample in there, and get it saturated with mushroom juice, really work it in there well. The more amatoxin is present the more there is to react and the better the test will perform in the event of a positive result.
Now add a couple of drops of concentrated hydrochloric acid solution (aq).
If the sample contains amatoxins, then a bluey-lilac-ish halo will appear around the sampled area, sometimes this can take several minutes, I would leave it to develop for 15 minutes at the absolute minimum, preferably double that for a good reliability, unless a result is shown earlier than this. If it does-THROW AWAY ANY AND ALL OF THE REST OF YOUR HARVEST! its not something I would like to do, would be pretty gutted if I had to toss the entire lot of a meal out. I have however once kept a batch of P.cyanescens, after a rust brown sporeprint developed (indicative of the possibility but by no means certainty of the presence of a Galerina, of which some are amatoxic, however this was a single, small mushroom (couple of centimeters wide post drying) amidst an entire large harvest of several hundred grams of definite cyans, and was weeded out. In fact believe I still have that mushroom somewhere actually)
Just a point!!! The amatoxin cyclopeptide core structure contains several rather unusual aminoacids, in a cyclic ring structure, linked to a center heterocyclic nucleus in the middle, the core is an indole molecule in the center. The Meixner test displays cross-reactivity with tryptamines, I.E psilocybin fungi. 100% crossreactivity for psilocin!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
I have wanted to test this for some time but have not been able due to the lack of an amatoxic mushroom to test with. Next time I find a deadly Lepiota or Amanita I shall have to bust out the lab. Oh, and take controls, people! Although i shouldn't have to tell anybody here that. IIRC the greenyblue-lilac-purplish color produced by a positive amatoxic substrate does not do this, whereas newspaper can go pink after a time in the sunlight, UV being responsible. Do the Meixner test out of bright sunlight if at all possible, and there is no reason it should not.
Do bear in mind also, that not all amatoxic species, all of the time, react. You are very unlikely, ever, ever to get a false positive, but some amatoxic species occasionally fruit a few that contain either extremely little, or perhaps even no amatoxin! particularly, I read, in the case of Amanita virosa, the destroying (or death) angel. (the species epithet coming from the latin 'virosus' meaning poisonous, same as the root origin of the words 'virulent' and 'virus')
Can anyone get the full text of this paper, I wont post it as a request in the request thread for it is far more appropriate if it is put here.
http://www.jstor.org/pss/3759568
Now onto the colorimetric species diagnostic reagent tests, families applicable in particular, are Amanita, Russula and Lactarius, although reagent tests are valuable in many more instances.
I will give the reagent, its preparation if needs be, and both the practical use of it, and a couple of representative color changes and the species they occur in.
HCl-concentrated HCl solution can be dropped into various fungi, its used for one, in the aforementioned Meixner test, or a mixture of HCl and para-DMAB (dimethylaminobenzaldehyde) will give a rapid blue/violet color change with a tryptamine containing mushroom. Again, due to that damnable indolic moiety in the cyclopeptide amatoxins/phallotoxins/virotoxins I need to see if this will crossreact with a known amatoxic species.
This is a variation on erlich's reagent/van urk, and these likewise can be used. There are quite a few variations on the erlichs/van urk reagent theme actually, involving H2SO4, ferrous sulfate (this alone reacts with some fungi bear in mind) and ferric chloride (again, quite probably so does this)
KOH or NaOH as moderately concentrated (10-15%) solutions are very common, very easily available reagents and can be kept in vials in a kit for field-testing specimens.
For instance its capable of differentiating Amanita virosa from its very, very close lookalike, although somewhat rarer, A.verna, both lethal amatoxin-packing species, both pure ghost-white, with white spores, a large ring and pronounced volva. The flesh (cap in particular) of A.virosa stains yellow when treated with KOH or NaOH whereas that of A.verna does not. A.ocreata does, whereas the white form of the deathcap sensu stricto, A.phalloides var.alba does not.
A particularly striking KOH/NaOH reaction is that of Hapilopilus rutilans, or H.nidulans, the purple dye polypores, these are dangerous poisonous species to eat (they turn woody and totally uneatable physically as they age, but when younger are softer), these cause a delayed neurotoxic encepalopathy if eaten, but are used to make dye, by reaction with the hydroxides, on which they turn a shocking purple.
Gives a dark ink-black, blacker than the heart of condoleeza rice, although a little less corrosive, when applied to the cap of certain Cortinarius species (webcaps, some of which contain a particularly insidious, very, very slowly acting nephrotoxin structurally related to paraquat)
Iron salts, particularly the ferrous sulfate and ferric chloride are of use IDing boletes (which have spongy, squishy pores, rather than gills), and Russula or Lactarius spp. giving in various cases, no change, or a change to various blues, greens, pinkish-reds, purples, yellow/yellowish greens depending on species.
Phenol as a 3-4% solution gives color changes, IIRC yellow in the case of certain Agaricus. Not too well up on the use of phenol for colorimetric testing of fungi.
With all these, test various parts, stem, cap, cut open stem/cap, gills.
A very important and useful reagent, although it will take a bit of work, is Melzer's, which is used on spores, rather than cap or stem, although quite possibly in some species, I don't know for sure would work on gill pieces.
The formula is, to make up 100ml, which will last a long time
dH2O: 100ml
Chloral hydrate: 100g
Iodine crystals: 2.5g
KI: 7.5g
This is used on spore print material, scraped onto glass (it can react with paper)
Reactions can be either none, amyloid, or dextrinoid.
Amyloid reaction is a dark violet-black. Same color as potato treated with tincture of iodine, dextrinoid is a rufous reddish-brown color.
One other thing, which I have been meaning to do, but haven't found time, is test things. ID species, test other chemicals on them. Plenty of books give a characteristic rxn or two, but not a comprehensive be-all-end-all-fuck-your-granny-in-the-process guide to the fungal anonymity apocalypse.
The intent being to have fun, have a good meal/trip and not to shit your liver out in liquid form.
When hunting, harvest by digging up the mushroom using a knife or spoon, do not yank them up by the stem, or rip the caps alone off, the stem, and base of the stem are vital diagnostic characteristics for telling what you have. Is a volva (cup-like structure) present at the base of the stem? this is diagnostic of two families, Amanita, and Volvariella, which are unrelated (confusion of the edible straw mushroom popular with orientals, and often used in chinese food, Volvariella spp. with deathcaps of varying species such as Amanita phalloides, A.ocreata and some of the similar looking Amanita subsection Phalloidea members is a major cause of death amongst eastasian immigrant mushroom hunters coming to the states or europe who make a dangerous fuckup as they look quite similar)
Be very wary and careful of any mushroom with a volva, do not bag said mushrooms in the same container you are taking any harvest that will be eaten home in unless it is to be fed to an enemy.
Is a ring present on the stem? in some species this tends to disintegrate or fray with age, and in some others, it is moveable up and down the stem if you do so carefully, IIRC, the parasol mushroom, Lepiota procera, a large member of the Lepiota family, which unlike some,which are deadly poisonous, is edible, and indeed, one I rather like to eat, can have its ring slid/rolled up and down the stem if you do it carefully. A few fungi have double rings, but this isn't particularly common.
Pay close attention to the morphology of the cap, and stem. Does the cap have a peelable surface, if the species has a cap. Some do not. Some species one can peel it off, such as the slippery jack bolete (suillus luteus), the related larch bolete (also in the genus Suillus), as do the death cap (Amanita phalloides) and common button mushrooms bought in stores, of the genus Agaricus, most commonly A.bitorquis (ick!....I don't like store bought mushrooms at all, apart from shiitake, oysters etc.) Whilst others do not, for example Mycena spp. and a good few of the Coprinus species. Does the cap become viscid when wet? or is it permanently sticky/glutinous, or is it not so at all? Note texture and smell of the fruitbody, it can help to crush some to release any smell more.
Some fungi have very diagnostic odours, such as in certain Russulas, the smell of decaying shellfish for R.foetens, in R.fellea, there is a pronounced smell of geranium, or in the case of, for example, Russula queletii, the scent of fresh apples.
Check gill structure (gills decurrent, running down the stem? adnexed? adnate? detatched from the stem? texture and resilience of gills, are they waxy? firm? do they break easily? pores like a bolete? some fungi, including a few absolutely excellent edibles, the hedgehog fungi, have pointy spines pointing downwards from the underside of the cap) Some fungi have no true gills, but instead, raised, forking, ridge-like pseudogills forming the fertile area, and some, have no gills at all, such as the horn of plenty/black funnel of death, known to be a very sought after edible, meant to be particularly good in soup or stew, after being dried for storage like one can with porcini/cep/Boletus edulis, but I have never been lucky enough to find any

In some genera, SOME, mind you, but not all, taste is important. Russula, Lactarius, and to a lesser extent, Boletus and their relatives such as Suillus and Chalciporus (C.piperatus for example, the peppery bolete, I use as a spice, dried and ground, just like I use chillis or pepper, its hot and firey, but in a different way to either spice)
The procedure for tasting is to break off a small piece of cap, and nibble it between the teeth for a short time, make sure it gets on the tongue, is it sweet? acrid? or is there a specific taste of something? In particular with Russula and Lactarius species, this test is borderline vital, and in these two families, there are none that a mere taste will kill. Although actually eating a scant handful of Lactarius species has done so in the past, and Russula subnigricans, the blackening russula (not to be confused with R.nigricans or R.nigrescens) contains a potent myotoxin, cycloprop-2-ene-1-carboxylic acid, causing severe rhabdomyolysis and subsequent kidney failure, this species can be lethal, and most painfully, slowly so at that.
Taste, but do not swallow. Spit.
Some fungi take time for a taste to develop, others start one way, then turn another, such as starting mild, but with a rapid, or slow development of an intense acrid burning sensation in the case of some of the Lactarius (milk caps).
In the case of this family, milk cap, comes from their bleeding a milky liquid substance when injured. Cause this to happen if you think you may have one from this genus, observe the consistency and color of the 'milk', some change color on exposure to air after a few minutes, blotting some milk onto white, absorbent paper such as a tissue, or bit of blotting paper can aid the development of the change. There are some good edibles to be had here, one or two species, such as L.torminosus, the wooly milk-cap may be fatal if eaten, but most likely will just cause severe GI irritation and accompanying miserable symptoms for a day or two. The toxins are most likely in most cases to be sesquiterpene compounds, as are those in many of the acrid Russulas (brittlegills). The Russulas, are, with the exception of the myotoxin-bearing R.subnigricans, not fatal, barring some freak ill luck, and besides, are so acrid in many cases for the toxic ones (in fact almost all toxic russilas, bar that species have a hot, burning taste. With very few exceptions, all burning russulas are for the chuck, and some consider it a general rule that all mild ones are edible, I do not rely on rules of thumb though to be exact, not when it comes to potential toxicity, but its very unlikely that you will be poisoned by a mild-tasting Russula.
There are some genera that you simply do not taste. Amanitas, don't do it. Inocybes, don't do it-there aren't any worth eating, and quite possibly none safe to eat either. The entire genus is chock-full of muscarine, with a few exceptions. Most are incredibly hard to ID, and impossible to do so without a microscope to check the spores, gill microstructure and hyphae out. Fun to pick for the challenge of an ID, but certainly not for eating. One mycologist became so ill after tasting a tiny piece of Amanita xanthocephala, an unusual species, not amatoxic, but most virulent nevertheless, that he broke his toes trying to get to the bathroom he was in such a hurry. And tasting a small piece of what turned out to be Podostroma cornu-damae, an oriental club fungus/coral type bugger would at the least burn and blister like mustard gas, and quite possibly prove fatal, although fungi of such extreme toxicity are the exception rather than the rule, and tasting even an amatoxic species by accident is very unlikely to kill you, although may nevertheless make you severely ill and cause liver damage to some degree.
Eat no mushroom that displays bioluminescence. All of them I am aware of that do phosphoresce in the dark, such as Lampteromyces japonicus, and Omphalotus species (the jack'o'lantern fungi) are poisonous. Interestingly the latter family produce their phosphorescence in the same way that fireflies do, the reaction between luciferin and luciferase. Eat something that glows in the dark, and you will regret it. Won't kill you, but will cause very severe GI upset.
Don't taste any Lepiota species either, many contain the same deadly amatoxins that the Amanita (subsection Phalloidea) do. Likewise for Galerina, and Conocybe, there are psilocybin-bearing members of both, but likewise amatoxin carrying ones, avoid tasting or eating these families in their entirety, its not worth repeatedly squirting your liver and kidneys down the inside of your trouser leg in a cloud of blood and cholera-esque chunky fecal soup until you shrivel up, turn yellow, and die over several weeks.
More on toxicity later, I'll give a breakdown of the main groups of toxic compounds, and the species (not totally comprehensive, its a bit too much to expect of me to know and to remember to name every single species in every family containing a given toxin or toxins, but I will give a wide range, of both the common ones, and a fair few more unusual and little nown nasties, where they are found and how they work)
Take note also of the habitat-is it growing on wood, if so, if you can tell, what kind of wood? growing on turds? or near, but not ON shit. In lawns/grassland? woodland? many fungi are what is called mycorrhizal, meaning they form an intimate composite structure between plant roots and the mushroom mycelium. If growing in association with plants, take samples of the nearby (for a few meters) trees and shrubs, most commonly a mycorrhizal fungus will form an association between a tree or woody shrub, macrofungi growing in association with herbs are much less common, and in the case of ergots, take at least one entire grass plant, leaving sclerotia in the seedhead for ID, as the host species often defines the parasite, C.purpurea excepted, which is unusual amongst the Clavicipitacea in being a bit of a whore really, where as others such as C.fusiformis are more picky, there is even one species specialising in bamboo.
Take spore prints, this is a very important step if you are uncertain of the identification. A microscope is helpful too, for some genera, it is vital if you are to ID down to species level, such as the genus Inocybe in particular (not that any of these are ones you want to eat, although a few contain psilocybin and psilocin, I.aeruginescens contains a novel tryptamine, the trimethyltryptammonium quat of psilocybin, although this will not pass the BBB, and is a POTENT 5HT6 agonist, which will make you feel very, very, very severely nauseated and cause quite spectacular vomiting, if its active in vivo at all, compare with ondansetron etc. the antinauseant drugs. The tryptamine bearing Inocybes in many cases contain very low concentrations of actives, avoid them IMO, as most species are full of the cholinergic neurotoxin muscarine, in many cases in large quantities, in some, enough to prove fatal) Pretty much all Inocybes are small to moderate sized LBMs (little brown mushrooms) with fibrous cap surfaces and very hard to ID.
Note spore color, and miscroscopic characteristics if you have a microscope. I need a new one :/
Next-Colorimetric reactions and reagent testing.
Testing fungi with chemicals is a very useful diagnostic tool, this can be done in the field in many cases.
First and foremost-the Meixner test. This isn't a test for species ID, but rather, for the presence of a specific group of toxins, the deadly cytotoxic amatoxins, on which, more information later on in my guide. It is sensitive to 2mcg of amatoxin (calc. as amatoxin-a, alpha-amanitin), a typical Amanita phalloides fruitbody of medium size contains between 4-7mg of total amatoxins, maybe a little more, maybe a little less, fungi are very variable as to chemotype, A.ocreata may contain even more. 10mg/fruitbody-1mg-gram wet tissue on average, or slightly more. 0.1mg/kg is more than enough to kill.
Take a small sample of mushroom cap, or if somebody has eaten the mushroom and an amatoxic species is suspected or even wondered about but only bits of stem or other waste is available, use that, and a slip of paper, this must be high in lignin, or the test will fail to work, or at best will work very poorly, indeed this has failed before, with the result of the direst consequences for the poor bastard the test had been done on behalf of when lab filter paper was used. Newspaper is an excellent type to use.
Mash the sample up good onto the paper, after drawing a pencil circle to tell you where the spot is going to appear if the test is positive, around the area. Work the sample in there, and get it saturated with mushroom juice, really work it in there well. The more amatoxin is present the more there is to react and the better the test will perform in the event of a positive result.
Now add a couple of drops of concentrated hydrochloric acid solution (aq).
If the sample contains amatoxins, then a bluey-lilac-ish halo will appear around the sampled area, sometimes this can take several minutes, I would leave it to develop for 15 minutes at the absolute minimum, preferably double that for a good reliability, unless a result is shown earlier than this. If it does-THROW AWAY ANY AND ALL OF THE REST OF YOUR HARVEST! its not something I would like to do, would be pretty gutted if I had to toss the entire lot of a meal out. I have however once kept a batch of P.cyanescens, after a rust brown sporeprint developed (indicative of the possibility but by no means certainty of the presence of a Galerina, of which some are amatoxic, however this was a single, small mushroom (couple of centimeters wide post drying) amidst an entire large harvest of several hundred grams of definite cyans, and was weeded out. In fact believe I still have that mushroom somewhere actually)
Just a point!!! The amatoxin cyclopeptide core structure contains several rather unusual aminoacids, in a cyclic ring structure, linked to a center heterocyclic nucleus in the middle, the core is an indole molecule in the center. The Meixner test displays cross-reactivity with tryptamines, I.E psilocybin fungi. 100% crossreactivity for psilocin!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
I have wanted to test this for some time but have not been able due to the lack of an amatoxic mushroom to test with. Next time I find a deadly Lepiota or Amanita I shall have to bust out the lab. Oh, and take controls, people! Although i shouldn't have to tell anybody here that. IIRC the greenyblue-lilac-purplish color produced by a positive amatoxic substrate does not do this, whereas newspaper can go pink after a time in the sunlight, UV being responsible. Do the Meixner test out of bright sunlight if at all possible, and there is no reason it should not.
Do bear in mind also, that not all amatoxic species, all of the time, react. You are very unlikely, ever, ever to get a false positive, but some amatoxic species occasionally fruit a few that contain either extremely little, or perhaps even no amatoxin! particularly, I read, in the case of Amanita virosa, the destroying (or death) angel. (the species epithet coming from the latin 'virosus' meaning poisonous, same as the root origin of the words 'virulent' and 'virus')
Can anyone get the full text of this paper, I wont post it as a request in the request thread for it is far more appropriate if it is put here.
http://www.jstor.org/pss/3759568
Now onto the colorimetric species diagnostic reagent tests, families applicable in particular, are Amanita, Russula and Lactarius, although reagent tests are valuable in many more instances.
I will give the reagent, its preparation if needs be, and both the practical use of it, and a couple of representative color changes and the species they occur in.
HCl-concentrated HCl solution can be dropped into various fungi, its used for one, in the aforementioned Meixner test, or a mixture of HCl and para-DMAB (dimethylaminobenzaldehyde) will give a rapid blue/violet color change with a tryptamine containing mushroom. Again, due to that damnable indolic moiety in the cyclopeptide amatoxins/phallotoxins/virotoxins I need to see if this will crossreact with a known amatoxic species.
This is a variation on erlich's reagent/van urk, and these likewise can be used. There are quite a few variations on the erlichs/van urk reagent theme actually, involving H2SO4, ferrous sulfate (this alone reacts with some fungi bear in mind) and ferric chloride (again, quite probably so does this)
KOH or NaOH as moderately concentrated (10-15%) solutions are very common, very easily available reagents and can be kept in vials in a kit for field-testing specimens.
For instance its capable of differentiating Amanita virosa from its very, very close lookalike, although somewhat rarer, A.verna, both lethal amatoxin-packing species, both pure ghost-white, with white spores, a large ring and pronounced volva. The flesh (cap in particular) of A.virosa stains yellow when treated with KOH or NaOH whereas that of A.verna does not. A.ocreata does, whereas the white form of the deathcap sensu stricto, A.phalloides var.alba does not.
A particularly striking KOH/NaOH reaction is that of Hapilopilus rutilans, or H.nidulans, the purple dye polypores, these are dangerous poisonous species to eat (they turn woody and totally uneatable physically as they age, but when younger are softer), these cause a delayed neurotoxic encepalopathy if eaten, but are used to make dye, by reaction with the hydroxides, on which they turn a shocking purple.
Gives a dark ink-black, blacker than the heart of condoleeza rice, although a little less corrosive, when applied to the cap of certain Cortinarius species (webcaps, some of which contain a particularly insidious, very, very slowly acting nephrotoxin structurally related to paraquat)
Iron salts, particularly the ferrous sulfate and ferric chloride are of use IDing boletes (which have spongy, squishy pores, rather than gills), and Russula or Lactarius spp. giving in various cases, no change, or a change to various blues, greens, pinkish-reds, purples, yellow/yellowish greens depending on species.
Phenol as a 3-4% solution gives color changes, IIRC yellow in the case of certain Agaricus. Not too well up on the use of phenol for colorimetric testing of fungi.
With all these, test various parts, stem, cap, cut open stem/cap, gills.
A very important and useful reagent, although it will take a bit of work, is Melzer's, which is used on spores, rather than cap or stem, although quite possibly in some species, I don't know for sure would work on gill pieces.
The formula is, to make up 100ml, which will last a long time
dH2O: 100ml
Chloral hydrate: 100g
Iodine crystals: 2.5g
KI: 7.5g
This is used on spore print material, scraped onto glass (it can react with paper)
Reactions can be either none, amyloid, or dextrinoid.
Amyloid reaction is a dark violet-black. Same color as potato treated with tincture of iodine, dextrinoid is a rufous reddish-brown color.
One other thing, which I have been meaning to do, but haven't found time, is test things. ID species, test other chemicals on them. Plenty of books give a characteristic rxn or two, but not a comprehensive be-all-end-all-fuck-your-granny-in-the-process guide to the fungal anonymity apocalypse.




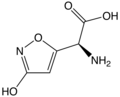
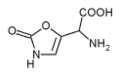
 )
)
 I will have to try and remember to do so next time I see them growing), I would guess of the gills also, this takes between ten and 15 minutes. 15-20% KOH or NaOH, orange discoloration of partial veil within 5-10 minutes, negative rxn to both FeSO4 and diluted H2SO4.
I will have to try and remember to do so next time I see them growing), I would guess of the gills also, this takes between ten and 15 minutes. 15-20% KOH or NaOH, orange discoloration of partial veil within 5-10 minutes, negative rxn to both FeSO4 and diluted H2SO4. )
)

 )
)
 A true morel in cross section, this is not Morchella conica, but another species, most likely Morchella esculenta.
A true morel in cross section, this is not Morchella conica, but another species, most likely Morchella esculenta.

 Ptychoverpa bohemica
Ptychoverpa bohemica The stipe of Ptychoverpa bohemica
The stipe of Ptychoverpa bohemica