Well considering no one else has reported success/failure retrying this method figured I'd give it a go.
Cu(C2H3O2)2 was prepared by double replacement reaction with Ca(C2H3O2)2 and Cu(SO4). 6.1g of tryptophan was dissolved in 600mL of warm water, to this a slight excess of the Cu(C2H3O2)2 was added. Immediately the solution turned purple and a precipitate rained down. After vacuum filtration and washing the compound was set aside.
75mL of DMSO was placed into a 250mL RBF. The chelate complex was there added. A liebig condenser was fitted onto the RBF and the solution was kept slightly under boiling temperature(with a little boiling in-between) for 12 minutes. The solution was allowed to cool and was filtered from elemental copper and some of the undissolved/unreacted complex.
The solution was diluted to 250mL in volume with D. H2O. An appropriate amount of NaOH solution was added to make the mixture basic. 35mL of toluene was stirred into the mixture 3 times and each time removed(not using a sep. funnel as there was a large amount of solid in the aq. layer *this is possibly a mistake, meaning the reaction may have been haulted early which is confusing because it was left hot and no more evolution of CO2 was noted for 2 minutes*).
The toluene had taken on an orange hue and was transferred to the sep. funnel where it was rinsed 3x with dilute HCl water. The toluene's color changed from orange to blue/yellow. A strange blue/black precipitate loomed between the organic and aq. layer, though the water was taken away with no visible contamination(slightly mint green tinge to the water). The pulls were basified with NaOH and again taken into Toluene which took on an orange hue.
Any suggestions on how to isolate the tryptamine specifically? I'm not afraid to run a column, just not sure what eluent to choose. Kind of wondering how much has possibly even been recovered. This route does not seem very appropriate compared to others around, it is being done simply out of inquiry and repeatability.
Cu(C2H3O2)2 was prepared by double replacement reaction with Ca(C2H3O2)2 and Cu(SO4). 6.1g of tryptophan was dissolved in 600mL of warm water, to this a slight excess of the Cu(C2H3O2)2 was added. Immediately the solution turned purple and a precipitate rained down. After vacuum filtration and washing the compound was set aside.
75mL of DMSO was placed into a 250mL RBF. The chelate complex was there added. A liebig condenser was fitted onto the RBF and the solution was kept slightly under boiling temperature(with a little boiling in-between) for 12 minutes. The solution was allowed to cool and was filtered from elemental copper and some of the undissolved/unreacted complex.
The solution was diluted to 250mL in volume with D. H2O. An appropriate amount of NaOH solution was added to make the mixture basic. 35mL of toluene was stirred into the mixture 3 times and each time removed(not using a sep. funnel as there was a large amount of solid in the aq. layer *this is possibly a mistake, meaning the reaction may have been haulted early which is confusing because it was left hot and no more evolution of CO2 was noted for 2 minutes*).
The toluene had taken on an orange hue and was transferred to the sep. funnel where it was rinsed 3x with dilute HCl water. The toluene's color changed from orange to blue/yellow. A strange blue/black precipitate loomed between the organic and aq. layer, though the water was taken away with no visible contamination(slightly mint green tinge to the water). The pulls were basified with NaOH and again taken into Toluene which took on an orange hue.
Any suggestions on how to isolate the tryptamine specifically? I'm not afraid to run a column, just not sure what eluent to choose. Kind of wondering how much has possibly even been recovered. This route does not seem very appropriate compared to others around, it is being done simply out of inquiry and repeatability.




 . Trying to salvage anything usable.
. Trying to salvage anything usable.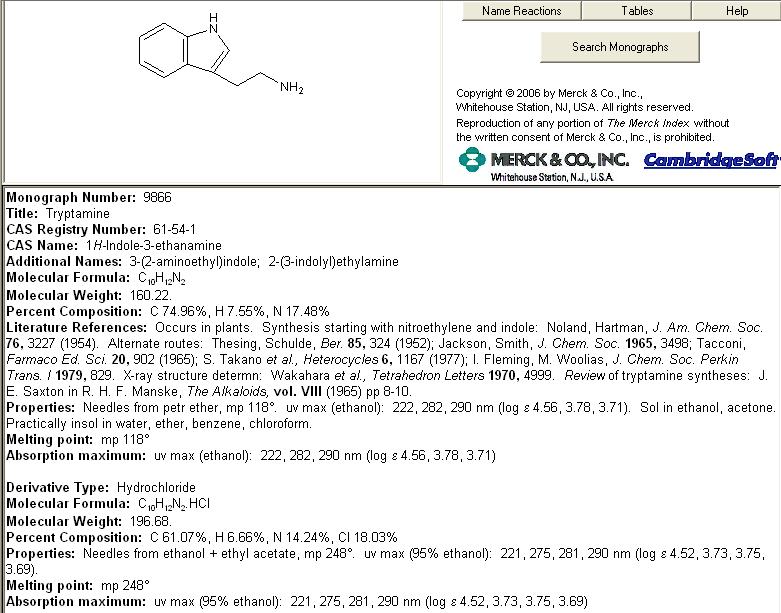


 ).
). .
.