Post by: Lego on September 18, 2004, 10:37:00 AM
Post 518711 (https://www.thevespiary.org/talk/index.php?topic=13048.msg51871100#msg51871100)
(Lego: "DMT from indigo in two steps?", Tryptamine Chemistry) Lego searched the literature in order to evaluate if it is possible to alkylate indoles in the 3 position. This would offer a one-step synthesis of tryptamines directly from indoles.The problem is the highly electron rich pyrrole ring. It can bee alkylated either at the 3-position (what we want), the 2-position (bad) and the 1-position (the nitrogen of the pyrrole, bad too). As the 3-position is the most activated position it should bee the prefered position of attack.
There are two imaginable ways of alkylating indoles, direct or with a catalyst (like a Friedel-Crafts alkylation).
The other choice is alkylating agent, it can bee either an alkyl halogenide or a alcohol.
Here are some examples of indole alkylations in the 3-position:
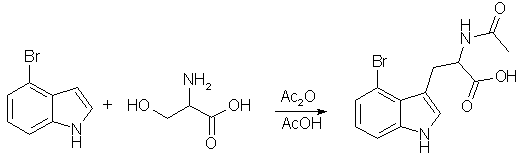
Heterocycles, 2001, 55(4), 653-660:
A mixture of DL-serine (1.20 g, 11.4 mmol) and acetic anhydride (2.4 mL, 23 mmol) in AcOH (7.0 mL) was heated for 1 h at 80 oC, and then 4-bromoindole (1.12 g, 5.70 mmol) was added to the solution. After being heated at 80 °C for 1.5 h, the reaction mixture was basified with 30% aqueous NaOH and washed three times with benzene-AcOEt (1 : 1). The aqueous layer was acidified with concentrated HCl and extracted three times with benzene-AcOEt (1 : 1). The organic layer was washed with brine and dried over MgSO4. After evaporation of the solvent, resulting crude amorphous solid (1.13 g) was subjected to chromatography over neutralized silica gel (benzene : AcOEt = 1 : 5 ~ 1 : 8) to give the acid {4-bromo-acetyl-tryptophan} (1.06 g, 57%) as a pale brown amorphous solid.
The authors prepared plenty of other substituted tryptophans with the same method (e.g. 7-bromo in 84% yield w/o chromatographic workup).
To avoid over-alkylation it is possible to use an excess of indole compared to alkylating agent or to block the 2-position e.g. by a carboxylic acid ester which can bee removed by decarboxylation,
Post 531852 (missing)
(Lego: "Hemetsberger-Knittel: Benzaldehydes to Indoles", Tryptamine Chemistry).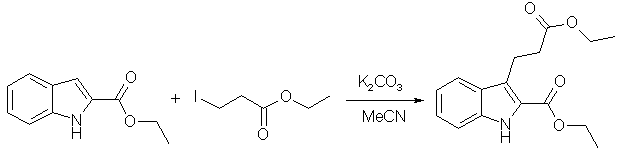
J. Med. Chem., 1991, 34(4), 1283-1292:
2-Carboxy-3-indolepropanoic Acid: Ethyl 2-indolecarboxylate (3 g, 15.9 mmol), ethyl 3-iodopropanoate (5.4 g, 22.3 mmol), K2CO3 (5 g), and acetonitrile (50 mL) were combined and heated to reflux for 48 h. The mixture was poured into H2O (50 mL) and the aqueous mixture was extracted with ether (3 x 75 mL). The combined ether solutions were washed with H2O (3 x 30 mL), dried (Na2SO4), and concentrated in vacuo. The resulting crude diester was hyrolyzed to diacid (2.37 g, 64%) as described.
If no strong base is used the alkylation of pyrrole nitrogen is very unlikely, therefore it should bee possible to alkylate indoles with either N,N-dimethyl-2-aminoethanol (cheap and commercial available) with the first procedure or with 2-chloro-N,N-dimethylethylamine (also cheap and commercial available).
Please not that the reaction is different than this one:
../rhodium/chemistry
/dmt.indole.grignard.html (https://www.thevespiary.org/rhodium/Rhodium/chemistry
/dmt.indole.grignard.html)
Lego remembers someone who was able to alkylate an excess of indole with a N-disubstituted 2-chloroethylamine in THF with ZnCl2 with very good yields and obtain the product by sublimation.
What do you think?
Post by: Xicori on September 21, 2004, 08:44:00 AM
Thank you, Lego for starting a thread on this very interesting topic - If the method turns out to be useful it would provide a 1-step-route to many N,N-disubstituted Tryptamines without the need of any highly watched chemicals.
2-(Dimethylamino) ethylchloride hydrochloride, 2-(Diethylamino) ethylchloride hydrochloride, and 2-(Diisopropylamino) ethylchloride hydrochloride are all cheap and commercially available, the drawback is that they are all rated fairly toxic.
Now there are some points to be discussed:
1. An excess of Indole is needed to prevent alkylation of the 2-position and on the indolic nitrogen. I think a 50% molar excess should be enough. What do you think?
2. Solvent. A wise choice seems to be THF. - It is inert to the reaction conditions, has a low boiling point and can be dried easily with the aid of molecular sieves.
3. reaction workup. Distilling off the remaining indole at reduced pressure and then distill/sublime the reaction product. What other methods come to mind (beside column chromatography)?
This is a hypothetical procedure for the synthesis of DIPT that might work fairly well:
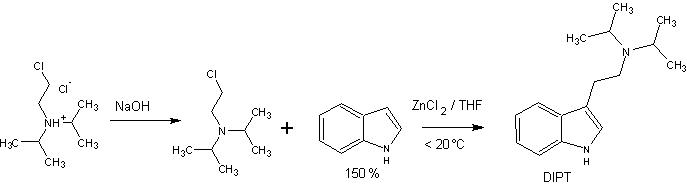
20g (0,1mol) of 2-(Diisopropylamino) ethylchloride hydrochloride are dissolved in 100ml water. The mixture is cooled, and under stirring a solution of 4,4g (0,11mol) NaOH in 15ml water is added dropwise. After finished addition the mixture is stirred for further 30min, and the solution is extracted twice with 50ml of Et2O. The Ether phases are combined and washed twice with brine. The ether is dried with Na2SO4, and the solvent evaporated tp yield the 2-(Diisopropylamino) ethylchloride freebase.
The freebase is taken up in 50ml THF and poured into an addition funnel, protected with a CaCl2 drying tube.
A flask, equipped with a reflux condensor, magnetic stirbar, drying tube and the above dropping funnel is charged with 17,6g (0,15mol) of Indole in 150ml THF, and 2,8g (0,02mol) of anhydrous ZnCl2. Under stirring the 2-(Diisopropylamino) ethylchloride is added dropwise, while cooling with an ice bath to keep the temperature low.
The mixture is stirred overnight and poured onto ice. If the pH is not <3 a few drops of HCl are added, and the solution is washed twice with Et2O, made strogly alkaline (until the Zn-salts dissolve again) and extracted twice with DCM. The DCM is washed with water and brine, dried over sodium sulfate and stripped off. The residue is distilled under a high vacuum to yield indole (b.p ~ 50°C @ 0,1mmHg), and the residue in the distilling flask is subjected to vacuum subblimation to yield DIPT freebase in a hopefully good yield!
I would be very pleased to get the impression and ideas of other bees. Let´s push things forward...
best wishes
xicori
Edit: Please could someone look up the physical properties of the 2-(Diisopropylamino) ethylchloride (base)? - I only have access to data of the hydrohloride salt.
Post by: Lego on September 21, 2004, 01:05:00 PM
Post 519153 (missing)
(Vitus_Verdegast: "Monitored for reagents used in chem weapons", Law and Order) this compound is a watched precursor in the EU!All N,N-dialkylated aminoethyl-2-chlorides or the corresponding ethanolamines are precursors for chemical warfare agents.
Bee careful!!!
According to Beilstein Crossfire:
Chemical Name: (2-chloro-ethyl)-diisopropyl-amine
(2-Chlor-aethyl)-diisopropyl-amin
2-chloro-N,N-diisopropylethylamine
Autoname: (2-chloro-ethyl)-diisopropyl-amine
CAS Registry Number: 96-79-7
Molecular Formula: C8H18ClN
Molecular Weight: 163.69
Melting Point:
118 - 1271 1
Ref. 1: Ortho Pharmaceutical Corp.;
Patent US2949485 (http://l2.espacenet.com/dips/viewer?PN=US2949485&CY=gb&LG=en&DB=EPD)
; 1960; Chem.Abstr.; EN; 55; 1448e; 1961.Boiling Point:
48 - 50°C (1 torr)1 1
66 - 67 (13 torr)2 1
Ref. 1: Vecchi; Melone; ANCRAI; Ann.Chim.(Rome); 49; 1959; 1192, 1203.
Ref. 2: Wright et al.; JACSAT; J.Amer.Chem.Soc.; 72; 1950; 3536,3537.
Post by: Rhodium on September 21, 2004, 02:06:00 PM
contained the preparation of ten different N,N-dialkylaminoethyl chlorides
as well as the corresponding N,N-dialkylaminoethanol precursors.
Histamine Antagonists. VII. Phenothiazine Derivatives
John B. Wright, Edward H. Lincoln, Richard V. Heinzelmann, James H. Hunter
J. Am. Chem. Soc. 72, 3536-3539 (1950)
(https://www.thevespiary.org/rhodium/Rhodium/hive/hiveboard/picproxie_imgs/pdf.gif)