"Tryptophan is isolated from casein in 0.7% yield by Cox and King's
[OS, 10, 100 (1930)] adaptation of the procedures given by Hopkins and Cole
[J. Physiol., 27, 418 (1901-02)], Dakin
[Biochem. J., 12, 290 (1918)] and Onslow
[Biochem. J., 15, 392 (1921)]. Commercial pancreatin [yes, from pancreas, easily extracted] is added to an aqueous suspension of commercial casein, [~3% of milk] Na
2CO
3 and NaF. The mixture is saturated with toluene and allowed to stand at 37° for 5 days. It is shaken daily during this period. Additional pancreatin is added and the mixture is allowed to stand for 12 days. The precipitate which forms on standing overnight in the fridge is reserved for the preparation of tyrosine. H
2SO
4 is added to the filtrate, the solution is cooled, a solution of HgSO
4 in H
2SO
4 is added, the mixture is allowed to stand for 48 hrs. and the supernatant liquid is siphoned.
The yellow residual material is washed thoroughly first with a H
2SO
4 solution of HgSO
4 to remove tyrosine and then with dH
2O to remove H
2SO
4. The moist precipitate is suspended in H
2O, a hot solution of Ba(OH)
2 is added until the mixture is alkaline to phenolphthalein and the Hg+2 removed as HgS. Ba+2 is removed as BaSO
4, the filtrate is evaporated
in vacuo and the aqueous solution is extracted repeatedly with
n-butanol. The butanol solution of tryptophan is distilled
in vacuo, the residual material is cooled and filtered, the crude tryptophan is dissolved in hot 60% EtOH, insoluble material is removed, the EtOH solution is decolorized with Norit and the purified tryptophan is washed with EtOH and Et
2O. Tryptophan has been isolated from various proteins by this method even though the isolation and purification procedures are long and tedious.
Sahyun
[US2416956, Texas Repts. Biol. Med., 7, 137 (1949)] has recently described a method for the large-scale preparation of crude tryptophan by selective adsorption on C."---
Max S. Dunn and Louis B. Rockland "Until recently, DL-tryptophan was synthesized exclusively from indole-3-aldehyde. Ellinger and Flamand
[Ber., 40, 3029 (1907), Ztschr. f. physio. Chem., 55, 8 (1909)] and Restelli
[Anales Soc. Quim. Argentina, 23, 58 (1945)] used the Erlenmeyer-Plochl synthesis. Majima
[Ber., 55, 3859 (1922)] used the hydantoin synthesis. He prepared the requisite aldehyde from indoleMgBr and ethyl formate.

Boyd and Robson
[Biochem. J., 29, 2256 (1935)] used a similar method to prepare this amino acid. The aldehyde was condensed with hydantoin in the presence of piperidine in 65% yield. Reduction with ammonium sulfide for 500 hrs. gave the amino acid in 70% yield.

The chief drawback in the above methods was the preparation of the aldehyde. Attention was directed toward improving the yield in the preparation of this compound. A synthesis from
o-nitrotoluene was recently described
[J. Chem. Soc., ?, 629 (1944)]. The Boyd-Robson synthesis was then followed, with considerable increase in overall yield. The
beta-indolemethylhydantoin was reduced catalytically in quantitative yield.

Recently it was found that indole-3-aldehyde may be prepared conveniently according to the following equation
[J. Am. Chem. Soc., 68, 1156 (1946)]:
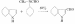
A modified Sorensen synthesis was employed by Snyder
[J. Am. Chem. Soc., 66, 350 (1944)] and Albertson
[J. Am. Chem. Soc., 66, 500 (1944)] and their coworkers. These authors discovered that gramine methiodide could be used as an alkylating agent. The reactions from indole are:
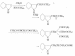
Albertson and coworkers
[J. Am. Chem. Soc., 67, 36 (1945)] found that if 2 moles of methyl sulfate is added to a mixture of 1 mole of gramine and 1 mole of ethylacetamidomalonate in alcoholic sodium ethoxide the yield in the alkylation step is 95%. When gramine was heated with ethyl acetamidomalonate in boiling xylene with powdered NaOH as catalyst, alkylation occured to the extent of 90%
[J. Am. Chem. Soc., 67, 38 (1945)].
Tryptophan was prepared in 71% overall yield from indole when ethyl acetamidocyanoacetate was substituted for the corresponding malonate
[J. Am. Chem. Soc., 67, 502 (1945)].
A variant of the above method was explored by Little and Weisblat
[J. Am. Chem. Soc., 69, 2118 (1947)]. They used ethyl nitroacetate in place of ethyl acetamidomalonate. The ester condensed with gramine to give ethyl
alpha-nitro-
beta-(3-indolyl)-propionate which was reduced and hydrolysed to the amino acid. The overall yield from gramine was 50%.
An interesting DL-tryptophan synthesis which did not use indole as the starting material was reported recently
[J. Am. Chem. Soc., 70, 2765 (1948)]. Acrolein was condensed with ethyl acetamidomalonate and the resulting aldehydo-ester isolated as the phenylhydrazone. The latter was subjected to a Fischer indole synthesis where upon the same ester resulted as obtained from gramine and ethyl acetamidomalonate. After hydrolysis and decarboxylation the amino acid was secured in 45% overall yield. The steps are:

A feature of this synthesis not shared by others is its adaptability to the preparation of nuclear substituted derivitaves of DL-tryptophan."---
S. Archer all of this was from
Amino Acids and Proteins: Theory, Methods, Application edited by
David Greenberg, 1951.
if any of these refs are ever looked up and found useful, posthumously thank halfapint. and thank Cornell for putting this excellent 950 page book on the web for free. i didn't think anyone would get interested if i just posted the link or the page files. the first part is an outline of the tryptophan from casein synth. that Rhodium has in detail. if you think it sucks, and yes i know it does, blame tryptophan.
http://historical.library.cornell.edu/cgi-bin/cul.neh/docviewer?did=greenber%5Bquote%5Dyou
can only see one page at a time, but you can jump to any page[/quote]