J.Med.Chem. - 24,8.1981;9404-allyloxy-3,5-dimethoxybenzaldehyde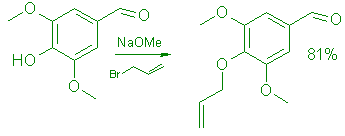
To 18.2g (0.1 mol) of syringic aldehyde in 40 mL of MeOH was added a solution of 6g (0.11 mol) of NaOMe in 35mL of MeOH. A clear yellow solution was formed. Allyl bromide (15.4g 0.13mol) was added, then the mixture was refluxed for 1.5h. A clear solution was formed within 15 min. The solvent was removed in vacuo and the residue recrystallized twice from hexane: yield 18g (81%)M mp 45-46°C
Alkylation with allyl tosylate should also be considered. TsO-Et=Me should be easy to prepare by the procedure outlined in
Post 492515
(Vitus_Verdegast: "Co(II) catalysed tosylation of alcohols", Chemistry Discourse).
PiHKAL #2 AL is a definite winner with the 3C analog yet to be tested.

J.Chem.Soc., Perkin Trans.2, 2000, 1119-11235-bromoveratraldehyde by solid phase bromination of veratraldehyde with NBS damn intresting article, lots of other aldehydes brominated.

Synthetic Communications 20;17;1990;2659-2666 - the original article can be found in
Post 514124 (not existing)5-Bromovanillin (5).
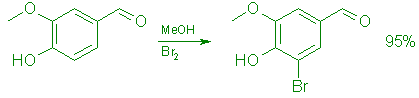
To a stirred, cooled (0°C) solution of 152.15 g (1.0 mol) of vanillin in 1.0 L of methanol was added during 20 min 176.0 g (1.1 mol) of bromine at such a rate that the temperature was kept below 20°C. The mixture was stirred at room temperature for 1 h, cooled to 0°C, and treated during 30 min with 500 mL of cold (5°C) water. Stirring was continued for 15 min and the product was collected by filtration. It was washed with water (4 x 500 mL), then with 500 mL of cold (0°C), 70% methanol, and dried in vacuo at 50°C overnight to give 218.5 g (95%) of 5 as pale yellow crystals, mp 163-164°C (Lit.,7 mp 163-164°C); UV (EtOH): 357 (E=4,200), 292 (e=9,800), 235 (e=15,300), 215 (e=19,000) nm; IR (CHCl3): 3300 (OH), 1675 (C=0) cm-1; 1H NMR (Me2SO-d6): S 3.93 (3 H, s), 7.35
(1 H, d, J=Hz), 7.60 (1 H, d, J=l Hz), 9.74 (1 H, s), and 10.51 (1 H, s, OH); MS m/z 230 (100, M+).
Anal. Calcd. for C8H7BrO3: C, 41.59; H, 3.05; Br, 34.59. Found: C, 41.48; H, 3.33; Br, 34.87.
Syringaldehyde (7) :
A: From 5-Bromovanillin (5).
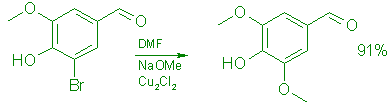
Under a blanket of argon, a solution of 214 g (0.93 mol) of 5-bromovanillin in 420 mL of anhydrous DMF was added during 25 min to a stirred slurry of freshly prepared, powdered sodium methoxide (3.72 mol, from 85.2 g of clean sodium in 1.0 L of CH3OH, followed by removal of the excess CH30H by distillation under vacuum) and 10.7 g (0.054 mol) of freshly prepared cuprous chloride in 150 mL of anhydrous DMF. The mixture was stirred at 97°C for 2.5 h, whereupon the color changed from blue to green after 30 min, and then to beige after 45 min. It was cooled to 60°C, and the DMF was evaporated under vacuum (0.2 Torr). To the residue was added 1.0 L of 15% brine and the mixture was stirred at 50°C for 30 min, cooled to 0°C and slowly acidified with 300 mL of cold (0°C) conc. hydrochloric acid. Stirring was continued at room temperature for 1.0 h and the crude product was collected by filtration. It was washed with 4 x 400 mL of cold H20 until neutral and extracted with warm (60°C) ethyl acetate (5 x 500 mL). The extract was evaporated to give 154 g (91%) of syringaldehyde, mp 109 111°C (t,it.,8 109-110°C); TLC (60:30:1 ethyl acetate:hexane:formic acid) showed only one spot, Rf 0.55; 5 had Rf 0.70. UV (EtOH): 365 (sh, e=1,200), 310 (e=13,100), 231 (e=15,980), 212 (E=18,500) nm; IR (CHCl3): 3520 (OH), 1685 (C=O); 1H NMR (CDC13 ): S 3.90 (6 H, s), 6.40 (1 H, s, OH), 7.10 (2 H, s), 9.75 (1 H, s); MS: m/z 182 (100, M+), 167 (12, M-CH3).
Anal. Calcd. for C9H1004: C, 59.34; H, 5.53. Found: C, 59.43; H, 5.28. Syringaldehyde prepared above may be crystallized from ethyl acetate (93% return) to give pale yellow crystals, mp 110-112°C.
B: From 3,5-Dibromo-4-hydroxybenzaldehyde (6).
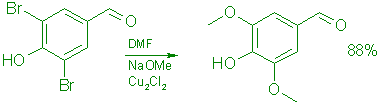
A 165-g (0.59 mol) batch of 6 in 156 mL of methanol and 312 mL of anhydrous DMF was reacted during 4.0 h with 4.78 moles of freshly prepared sodium methoxide in the presence of 9.45 g (0.047 mol) of cuprous chloride under the conditions described previously for the conversion of 5 to give 153.3 g (88% yield) of 7, mp 105-108°C.
3,4-Dibromo-4-hydroxybenzaldehyde:
A: From 4-Hydroxybenzaldehyde.
To a stirred, cooled (0°C) solution of 122.0 g (1.0 mol) 4-hydroxybenzaldehyde in 1.0 L of methanol was added during 30 min 325.0 g (2.2 mol) of bromine at such a rate that the temperature was kept below 20°C. The mixture was stirred at room temperature for 1.0 h, 800 mL of methanol was removed by distillation (water aspirator) at 50°C, and to the warm (45°C) solution was added 2.0 L of water during 20 min. The mixture was stirred at 0°C for 1.0 h and the product was collected by filtration. It was washed with water (5 x 1.0 L) and then with 500 mL of cold, 30% aqueous methanol, dried in vacuo at 70°C for 18 h to give 264.4 g (95.5%) of 6 as a colorless powder, mp 180-182°C. TLC (silica gel, 30:60:1 ethyl acetate: hexane: formic acid) showed only one spot under short wavelength UV light. UV (EtOH): 346 (E=8,000), 278 (E=9,000), and 221 (e=18,600) nm; IR (CHCl3): 3335, 1675, 1506, and 855 cm-1; 1H NMR (Me2SOd6) S 7.96 (2 H, s), 9.75 (1 H, s), and 10.23 (1 H, br, OH); MS: m/z 280 (M+). Anal. Calcd. for C7H4Br2O2: C, 30.04; H, 1.44; Br, 57.09. Found: C, 30.14; H, 1.49; Br, 56.94.
B: From p-Cresol.
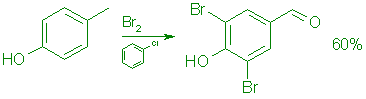
A cooled (10°C), stirred solution of 108.1 g (1.0 mol) of pcresol in 500 mL of chlorobenzene was treated during 30 min with 720 g (4.55 moles) of bromine in 600 mL of chlorobenzene at such a rate that the temperature was kept below 25°C. The mixture was then stirred at reflux for 4.5 h and concentrated in vacuo at 60°C to give 219 g of a red oil. This was dissolved in 500 mL of methanol, cooled to 5°C, and treated with 500 mL of 1.ON hydrochloric acid. The mixture was stirred at 5°C for 4.5 h, diluted with 1.0 L of cold water, and the product was collected by filtration. It was washed with 500 mL of cold 50% aqueous methanol and dried in vacuo at 35°C overnight to give 168.3 g (60%) of 6, mp 179-182°C.
UV (EtOH): 346 (e=8,000), 278 (e=9,000), and 220 (e=19,100) nm; 1H NMR (CDC13): S 6.43 (1 H, s, OH), 7.89 (2H, s), and 9.75 (1 H, s); MS: m/z 280 (100, M+).
Anal. Calcd for C7H4Br2O2: C, 30.04; H, 1.44; Br, 57.09. Found: C, 30.27; H, 1.65; Br, 56.94.
3,4,5-Trimethoxybenzaldehyde (1).

A stirred mixture of 153.0 g (0.84 mol) of syringaldehyde (7) in 1.0 L of acetone, was treated with 127.0 g (1.02 mol) of dimethyl sulfate, 118.0 g (1.11 mol) of anhyd. Na2CO3, and 3.0 g (0.053 mol) of KOH in 32 mL of water. The mixture was stirred under reflux for 18 h. cooled to room temperature and filtered. The filter cake was washed with acetone (3 x 200 mL) and the filtrate and washings were concentrated to ca. 200 mL. The stirred solution was diluted with 500 mL of water during 30 min, followed by 1.5 L of water during 15 min. Stirring was continued at room temperature for 15 min and then at 0°C for 30 min, and the product was collected by filtration. It was washed with water (4 x 2.0 L) and dried in vacuo at 25°C for 24 h to give 150.2 (91.5%) of 1 as colorless crystals, mp 73-75°C (Lit.,9 74°C). TLC (4:1 ether-hexane) revealed only one spot, Rf 0.57. UV (EtOH): 218 (e=25,400) and 287 (E=10,450) nm; IR (CHCl3): 1695 cm-1; 1H NMR (CDC13): S 3.89 (9 H, s), 7.09 (2 H, s), 9.81 (1 H, s); 13C (CDC13) S 56.07 (2 C, q, 2 x OCH3), 60.74 (1 H, q, OCH3), 106.55 (2 C, d, 2 x C=CH), 132.1 (1 C, s, ArC), 143.8 (1 C, s, ArC), 154.0 (2 C, s, 2 x ArC), 190.82 (1 C, d, CHO); MS: m/z 196 (M+).
Anal. Calcd. for C1OH1204: C, 61.22; H, 6.17. Found: C, 61.27; H, 6.24.
--
psyloxy--