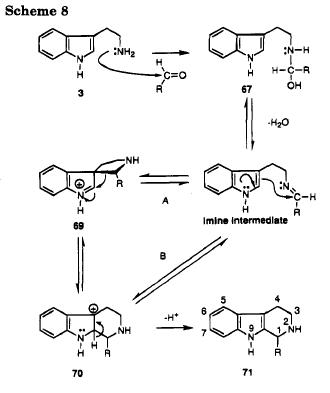
First, the mechanism needs to be considered.
"The Pictet-Spengler reaction has generally been thought to proceed via a spiroindolenine intermediate as shown in Scheme 8 (path A), although Casnatiso has shown that cyclization can occur by direct attack at position 2 of the indole (path B) when very reactive electrophiles are employed."
Therefore, the more the dipole moment is shifted toward the nitrogen, the more susceptible the carbon is to nucleophilic attack. The pKas of the imines are an indirect measure of electrophilic nature of the end of the imine containing the carbon. A Lewis acid is an electron acceptor; since the tryptophan methyl ester Schiff base has the lower pKa, it is more acidic and draws electron density away from the carbon, increasing its susceptibility to nucleophilic attack relative the the tryptamine Schiff base.
"These results can be rationalized by examination of the PKa values of the two [Schiff] bases: tryptamine, PKa = 10.2, tryptophan methyl ester, pKa = 7.29. The tryptophan methyl ester imine intermediate 19 is clearly more electrophilic...."
 Here is the the short answer to your question, antibody2.
Here is the the short answer to your question, antibody2. When the Schiff base of a secondary amine is formed (as in the case of the N-methyl tryptamine Schiff base) or an imine is protonated, the nitrogen receives a positive formal charge. This moeity is called an
iminium instead of an imine. Because the nitrogen is forced donate more than its desired share of electrons, its electrophilicity increases, and it draws electron density toward itself to an even greater extent than in the case of the tryptophan methyl ester Schiff base.
When I said "cannot be methylated," I was probably speaking too strongly, because certain rxn conditions could limit the formation of the of unwanted cyclic product. For instance, the reason that STAB (edit: which apparently cannot be used on unprotected indoles) is used is because the reagent is not strong enough to quickly reduce the aldehyde or ketone, yet as soon as the iminium is formed, the hydride attacks the electrophilic carbon before the ring can, preempting cyclization.








