An other way to indole 3 acetic acid:
Normal indole is an available chemical. At us, it is sold relative cheap (100g/25 euro). From this compound there are a lot of ways to get indole 3 acetic aciid what is the direct precursor of the DMT. Let's see an easy way:
Normal indole with diazoacetic acid ethyl ester in boiling ether gives indole 3 acetic acid ethyl ester. The diazoacetic acid loses the diazo part of the molecule and it just get's on the indole. The formation of the IAA ethyl ester is almost near to 90%. The reaction was described by Jackson in 1935.
The precusors are not hard to get. As I wrote, the indole is available in chem. supply shops. The diazo acetic acid ethyl ester can be made from glycine.
Synthesis of diazo acetic acid ethyl ester:
 from
from 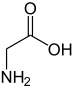
The glycine ethyl ester HCl is made by adding glycine to EtOH and bubbling dry HCl gas through it at 65-70 Celsius. The EtOH is also the solvent, so use more that the calculated amount. EtOH slould be added again and again to the reaction, because it will boil out from the mixture. You can get a recipe here: http://www.jbc.org/content/175/2/531.full.pdf
The ethyl ester should be diazotated with NaNO2. This part of the reaction is not so hard. Just cool down the chemicals, get a solvent what dissolves both ot the components (the NaNO2 and the glycine ethyl ester HCl). The glycine ethy ester HCl is soluble in ethanol, methanol, ether, benzne. Maybe the best would be to use methanol as a solvent (sodium-nitrite solubility in methanol: 4.4 g / 100 g 20°C). The diazo acetic acid ethyl ester is a yellow oily liquid. -22 Celsius melting point, boiling point, 140-141 Celsius.
The glycine ethyl ester is soluble in ether so just dissolve it in ether, add the indole to it and start to boil it. After the calculated amount of N2 evolved you are ready with the indole 3 acetic acid ethyl ester. Now you should just add the dimethyl amine to the mixture. And hydrogenate it to get the pure DMT.
-Evilblaze
Normal indole is an available chemical. At us, it is sold relative cheap (100g/25 euro). From this compound there are a lot of ways to get indole 3 acetic aciid what is the direct precursor of the DMT. Let's see an easy way:
Normal indole with diazoacetic acid ethyl ester in boiling ether gives indole 3 acetic acid ethyl ester. The diazoacetic acid loses the diazo part of the molecule and it just get's on the indole. The formation of the IAA ethyl ester is almost near to 90%. The reaction was described by Jackson in 1935.
The precusors are not hard to get. As I wrote, the indole is available in chem. supply shops. The diazo acetic acid ethyl ester can be made from glycine.
Synthesis of diazo acetic acid ethyl ester:
 from
from 
The glycine ethyl ester HCl is made by adding glycine to EtOH and bubbling dry HCl gas through it at 65-70 Celsius. The EtOH is also the solvent, so use more that the calculated amount. EtOH slould be added again and again to the reaction, because it will boil out from the mixture. You can get a recipe here: http://www.jbc.org/content/175/2/531.full.pdf
The ethyl ester should be diazotated with NaNO2. This part of the reaction is not so hard. Just cool down the chemicals, get a solvent what dissolves both ot the components (the NaNO2 and the glycine ethyl ester HCl). The glycine ethy ester HCl is soluble in ethanol, methanol, ether, benzne. Maybe the best would be to use methanol as a solvent (sodium-nitrite solubility in methanol: 4.4 g / 100 g 20°C). The diazo acetic acid ethyl ester is a yellow oily liquid. -22 Celsius melting point, boiling point, 140-141 Celsius.
The glycine ethyl ester is soluble in ether so just dissolve it in ether, add the indole to it and start to boil it. After the calculated amount of N2 evolved you are ready with the indole 3 acetic acid ethyl ester. Now you should just add the dimethyl amine to the mixture. And hydrogenate it to get the pure DMT.
-Evilblaze








