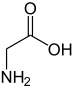
I thought I had my own way of carrying out the synthesis but then i realized the stabilized chloro dimethyl ammonium salt i wanted to add was insoluble in nonpolar solvents.
I thought i could use an IAA stabilised trianion (formed by reaction of IAA with BuLi) as nucleophile [Coupling reactions of indole-3-acetic acid derivatives. Synthesis of arcyriaflavin A Jan Bergman, Eva Koch and Benjamin Pelcman] add on the dimethyl amine followed by aqueous work up (think that would only work with an acid chloride derivative though)
I also gave michael addition a look but the alkyl chain is one kink too long :-)
Anyway heres a great paper using TCT as catalyst for solvent free michael reactions of indoles and pyroles. Enjoy!
SOme of you may recall the paper on TCT as efficient catalyst for turning alcohols in alkyl chlorides...I think I found that here.
I thought i could use an IAA stabilised trianion (formed by reaction of IAA with BuLi) as nucleophile [Coupling reactions of indole-3-acetic acid derivatives. Synthesis of arcyriaflavin A Jan Bergman, Eva Koch and Benjamin Pelcman] add on the dimethyl amine followed by aqueous work up (think that would only work with an acid chloride derivative though)
I also gave michael addition a look but the alkyl chain is one kink too long :-)
Anyway heres a great paper using TCT as catalyst for solvent free michael reactions of indoles and pyroles. Enjoy!
SOme of you may recall the paper on TCT as efficient catalyst for turning alcohols in alkyl chlorides...I think I found that here.





 from
from