Quote
Maybe just rent a helium tank from a party supply store?
They add oxygen so the little suckers don't pass out.. just an FYI





Maybe just rent a helium tank from a party supply store?






















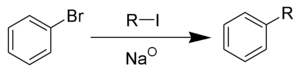
The acidity of carboxylic acids and 1º & 2º-amides acts to convert Grignard and alkyl lithium reagents to hydrocarbons (see equations), so these functional groups should be avoided when these reagents are used.





































 Members of Hyperlab have successfully used hydrocarbons such as propane, but it's inherently more dangerous because it's flammable
Members of Hyperlab have successfully used hydrocarbons such as propane, but it's inherently more dangerous because it's flammable  Helium is inert but not very dense
Helium is inert but not very dense  Nitrogen can be made from compressed air:
Nitrogen can be made from compressed air: